Cell Therapy Market Size, Share & Trends Analysis Report by Therapy Type (Autologous Cell Therapy, Allogeneic Cell Therapy), Cell Type, Technology, Therapy Technique, Cell Source, Application, End User and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2025–2035
|
Market Structure & Evolution |
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Cell Therapy Market Size, Share, and Growth
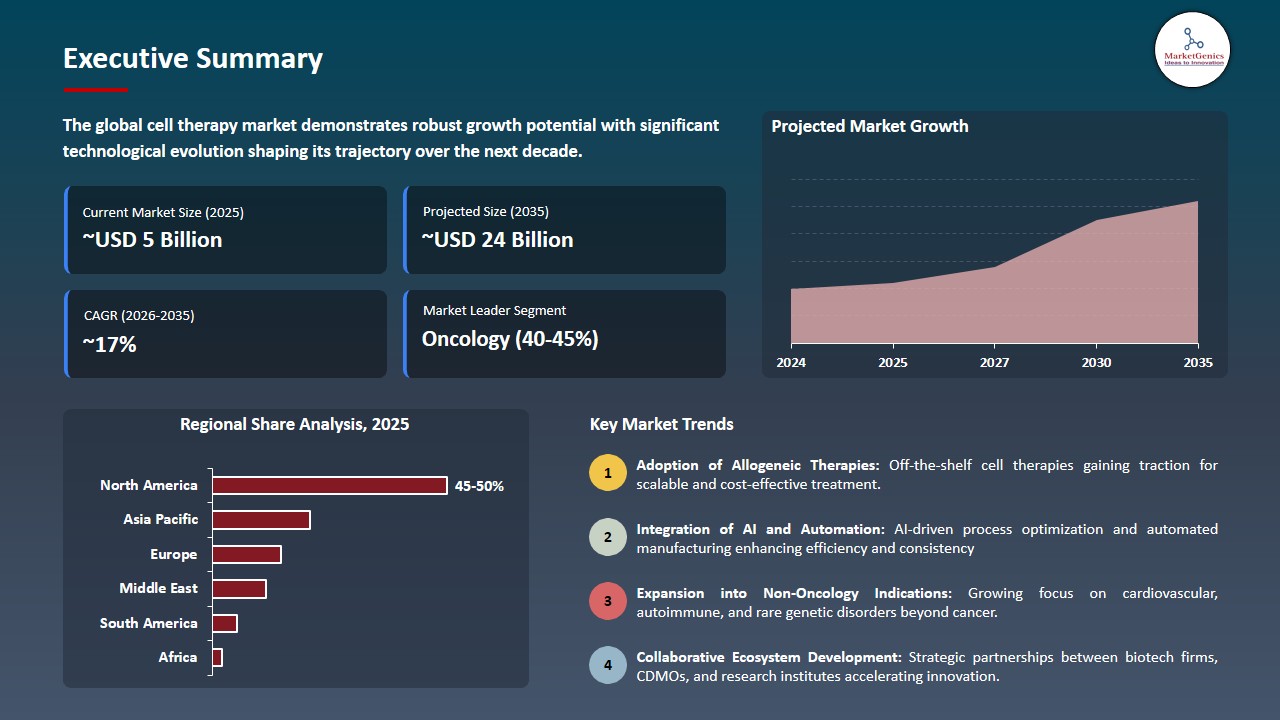
The global cell therapy market is experiencing robust growth, with its estimated value of USD 5.3 billion in the year 2025 and USD 24.4 billion by 2035, registering a CAGR of 16.5% during the forecast period.
Andreas Knaack the President of Invetech explained that their company developed predictive artificial intelligence models which they integrated into development and manufacturing processes to help cell therapy developers discover vital process parameters before time while they decreased production variability and expedited their journey toward creating patient-ready therapies.
The worldwide cell therapy market experiences fast growth because new technologies provide better medical solutions which people now demand. Thermo Fisher Scientific launched its advanced modular closed manufacturing systems in 2025 which automate essential processes for viral vector production while decreasing contamination risks and enabling companies to produce products at a commercial scale.
The increasing occurrence of cancer together with chronic diseases drives the demand for cell-based therapies which leads to funding for better manufacturing processes and advanced clinical research. The US FDA and EMA regulatory agencies provide market growth support through their established approval processes which help investors make informed decisions. The combination of technological advancement and strategic partnerships and regulatory frameworks provide patients with better treatment options.
The company provides multiple opportunities through its automated bioprocessing platforms and decentralized point-of-care manufacturing and viral vector production services and quality control automation and off-the-shelf allogeneic therapies.
The manufacturing process benefits from these areas because they help producers achieve better operational results while delivering consistent medical treatments and increasing their income throughout the regenerative medicine field. The cell therapy market will maintain its long-term growth through this integrated growth strategy which will also enhance worldwide patient health results and safety standards.

Cell Therapy Market Dynamics and Trends
Driver: Increasing Regulatory Backing Driving Adoption of Advanced Cell Therapies
-
Owing to flexible regulations along with expedited approvals like the Fast Track designation process and RMAT (Regenerative Medicine Advanced Therapy) infrastructure to help expedite the commercialization of next-generation therapies, the cell therapy industry has been experiencing healthy growth and attracting large amounts of funding.
- In 2025, India's Bharat Biotech opened a new subsidiary called Nucelion Therapeutics that specializes in developing cell and gene therapies, and operates in accordance with Good Manufacturing Practices (GMP), which ensures compliance with rigorous global regulations.
- Additionaly, to increasing numbers of development programs for treatment options for cancer, autoimmune disease & rare diseases, there is an accelerated rate of clinical trial activity due to increased flexibility in regulatory oversight. All these factors are likely to continue to escalate the growth of the cell therapy market.
Restraint: Complex Manufacturing and High Adoption Costs Limiting Cell Therapies
-
Complex manufacturing processes for cell therapies require a unique facility, trained personnel, thorough quality control, and continuous monitoring, creating barriers to therapy scale-up and cost-effective manufacture.
- The expensive price of therapy and lack of reimbursement will continue to restrict clinical adoption of this technology and slow down the transition from research to commercial use. The implementation complexity and up-front cost of integrating IoT-enabled sensors and real-time quality control technologies into bioreactors.
- For example, is a challenging justification for many developers, even though Industry 4.0 bioprocessing advancements are available. All these elements are expected to restrict the expansion of the cell therapy market.
Opportunity: Expansion into Emerging Diseases and Markets Expanding Cell Therapies Awareness
- Besides being applied for use in oncology/rare genetic disorder, currently, cell therapies are also being advanced in other types of diseases as well.
- For instance, collaborations such as the strategic agreement established between the Biotechnology Industry Research Assistance Council (BIRAC) of India and Miltenyi Biotec India, have been developed to improve local clinical research and manufacturing capabilities to support the wider application of cell therapies to meet unfulfilled medical needs.
- Emerging markets in the Asia Pacific region will also strengthen their cell therapy ecosystems through expanded trial activity, expanded investment into infrastructure, and establishing additional global partnerships providing opportunities for greater access, lower costs, and the ability to attract worldwide cell therapy companies. And thus, is expected to create more opportunities in future for cell therapy market.
Key Trend: AI, Digital Manufacturing, and Allogeneic Therapies
-
Cell therapies manufactured using industrialization methods are on the rise, with allogeneic or “off-the-shelf” therapies being developed much faster than autologous or “personalized” therapies. Nearly 60 allogeneic cell therapies have reached advanced development stages, and many biomanufacturers are increasing capacity to meet demand for large volumes of these products.
- Automation, artificial intelligence and machine learning, and the implementation of advanced systems are key topics at Industry Conferences and Summits; these technologies will help simplify the production processes, improve product quality, and decrease complexity in manufacturing. Therefore, is expected to influence significant trends in the cell therapy market.
Cell Therapy Market Analysis and Segmental Data
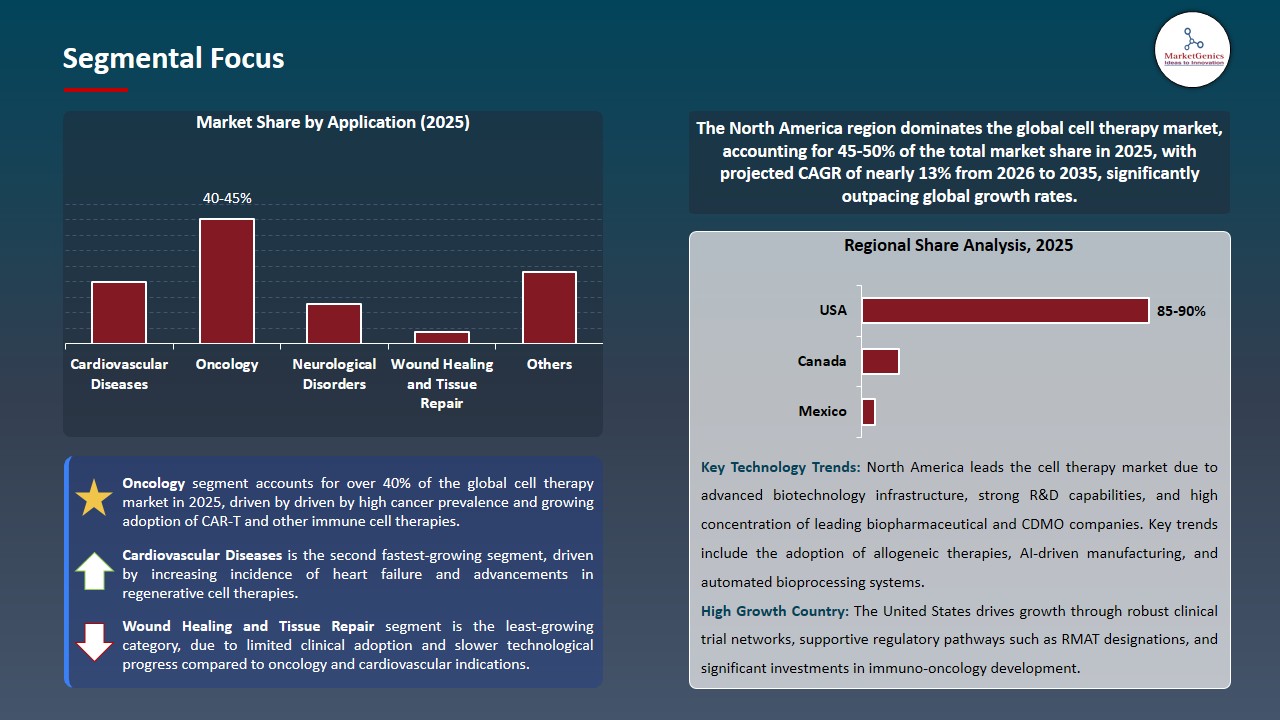
Oncology Dominates Global Cell Therapy Market Amid Rising Cancer Burden and Personalized Treatment Adoption
-
The global cell therapy market is dominated by the oncology segment because cancer shows high worldwide prevalence and there exists an urgent requirement for better treatments and CAR‑T and TCR and other personalized immune cell therapies show rapid development. The clinical and commercial advancement of CAR‑T personalized autologous treatments has shown better results for treating refractory blood cancers which medical researchers are now testing on solid tumor patients.
- Notably, the Chinese biotech company Carsgen Therapeutics announced its successful phase 2 trial results for stomach cancer treatment using its autologous CAR‑T product satri‑cel in 2025 yet the company now waits for regulatory authorities to complete their priority review process because oncology treats cancer patients who drive growth in cell therapy treatments.
- The expansion of oncology pipelines progresses faster because of strong financial backing and partnerships between different companies in the industry making it the leading segment within cell therapy market.
North America Dominates Cell Therapy Market Amid Advanced Infrastructure and Favorable Regulatory Support
-
North America leads the global cell therapy market because its strong biotechnology sector and advanced healthcare system and active regulatory system create an environment that supports research and product development. The United States benefits from strong research funding, high clinical trial activity, and the presence of major biopharmaceutical companies that drive adoption of cutting‑edge therapies.
- The U.S. Food and Drug Administration (FDA) together with other regulatory bodies provides expedited approval methods that allow drug developers to create new cell therapies through an efficient development process.
- The recent progress which includes U.S. biotech CDMOs establishing new manufacturing operations from preclinical research through to commercial production shows how both industry needs and manufacturing capacity have increased. High research investment together with a positive reimbursement system establishes North America as the leading force in the worldwide cell therapy market.
Cell Therapy Market Ecosystem
The global cell therapy market shows moderate consolidation during its development phase, while its commercial segment remains dominated by Gilead Sciences and Bristol Myers Squibb who operate as Tier 1 companies and Tier 2 and Tier 3 companies who develop specialized technologies.
The primary value chain components which determine operational expenses and operational abilities include clinical R&D and scalable manufacturing and CDMO services. Eli Lillys USD 2.4 billion purchase of Orna Therapeutics demonstrates the company’s strategic investment in in vivo cell therapy platforms to enhance its research and development capabilities.

Recent Development and Strategic Overview:
-
In August 2025, Adva Biotechnology and Autolomous established the Digital Pioneer Program to create a digital decentralized system for autologous cell therapy production through their automated manufacturing system which incorporates digital process management and complete production tracking for better operational efficiency and regulatory adherence during the entire therapy development process.
- In January 2026, Cellistic introduced three new iPSC-based GMP manufacturing platforms which include Echo-T and Echo-Cardio and Echo-Endothelial to support the development of scalable cell therapies for immuno-oncology and regenerative medicine while showing the industry's need for standardized production methods which produce ready-made therapeutic solutions.
Report Scope
|
Attribute |
Detail |
|
Market Size in 2025 |
USD 5.3 Bn |
|
Market Forecast Value in 2035 |
USD 24.4 Bn |
|
Growth Rate (CAGR) |
16.5% |
|
Forecast Period |
2025 – 2035 |
|
Historical Data Available for |
2020 – 2024 |
|
Market Size Units |
USD Billion for Value |
|
Report Format |
Electronic (PDF) + Excel |
|
Regions and Countries Covered |
|||||
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Cell Therapy Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Cell Therapy Market, By Therapy Type |
|
|
Cell Therapy Market, By Cell Type |
|
|
Cell Therapy Market, By Technology |
|
|
Cell Therapy Market, By Therapy Technique |
|
|
Cell Therapy Market, By Cell Source |
|
|
Cell Therapy Market, By Application |
|
|
Cell Therapy Market, By End User |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Natures
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Natures
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Cell Therapy Market Outlook
- 2.1.1. Cell Therapy Market Size (Value - US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Cell Therapy Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Ecosystem Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising prevalence of cancer and chronic diseases fueling demand for advanced therapies.
- 4.1.1.2. Technological innovations in CAR‑T, TCR, and allogeneic cell therapies enhancing efficacy and accessibility.
- 4.1.1.3. Supportive regulatory frameworks and expedited approval pathways accelerating development and commercialization.
- 4.1.2. Restraints
- 4.1.2.1. High manufacturing complexity and costs limiting large-scale adoption.
- 4.1.2.2. Stringent quality control and compliance requirements slowing production and scalability.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.5. Porter’s Five Forces Analysis
- 4.6. PESTEL Analysis
- 4.7. Global Cell Therapy Market Demand
- 4.7.1. Historical Market Size – Value (US$ Bn), 2020-2024
- 4.7.2. Current and Future Market Size – Value (US$ Bn), 2026–2035
- 4.7.2.1. Y-o-Y Growth Trends
- 4.7.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Cell Therapy Market Analysis, by Therapy Type
- 6.1. Key Segment Analysis
- 6.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Therapy Type, 2021-2035
- 6.2.1. Autologous Cell Therapy
- 6.2.2. Allogeneic Cell Therapy
- 7. Global Cell Therapy Market Analysis, by Cell Type
- 7.1. Key Segment Analysis
- 7.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Cell Type, 2021-2035
- 7.2.1. Stem Cell Therapy
- 7.2.1.1. Hematopoietic Stem Cells
- 7.2.1.2. Mesenchymal Stem Cells
- 7.2.1.3. Embryonic Stem Cells
- 7.2.1.4. Induced Pluripotent Stem Cells (iPSCs)
- 7.2.2. Immune Cell Therapy
- 7.2.2.1. T-Cell Therapy
- 7.2.2.2. CAR-T Cell Therapy
- 7.2.2.3. Natural Killer (NK) Cell Therapy
- 7.2.2.4. Dendritic Cell Therapy
- 7.2.3. Other Cell Therapies
- 7.2.3.1. Fibroblast-Based Therapies
- 7.2.3.2. Chondrocyte-Based Therapies
- 7.2.1. Stem Cell Therapy
- 8. Global Cell Therapy Market Analysis, by Technology
- 8.1. Key Segment Analysis
- 8.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Technology, 2021-2035
- 8.2.1. Somatic Cell Technology
- 8.2.2. Genome Editing Technology
- 8.2.3. Viral Vector Technology
- 8.2.4. Cell Immortalization Technology
- 8.2.5. Cell Plasticity Technology
- 8.2.6. 3D Cell Culture Technology
- 8.2.7. Others
- 9. Global Cell Therapy Market Analysis, by Therapy Technique
- 9.1. Key Segment Analysis
- 9.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Therapy Technique, 2021-2035
- 9.2.1. Stem Cell Therapy
- 9.2.2. Adoptive Cell Transfer (ACT)
- 9.2.2.1. CAR-T Cell Therapy
- 9.2.2.2. T-Cell Receptor (TCR) Therapy
- 9.2.2.3. Natural Killer (NK) Cell Therapy
- 9.2.3. Cell Vaccine Therapy
- 9.2.4. Fibroblast Cell Therapy
- 9.2.5. Chondrocyte Cell Therapy
- 9.2.6. Others
- 10. Global Cell Therapy Market Analysis, by Cell Source
- 10.1. Key Segment Analysis
- 10.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Cell Source, 2021-2035
- 10.2.1. Bone Marrow-Derived Cells
- 10.2.2. Umbilical Cord Blood-Derived Cells
- 10.2.3. Adipose Tissue-Derived Cells
- 10.2.4. Placenta-Derived Cells
- 10.2.5. Induced Pluripotent Stem Cells (iPSCs)
- 10.2.6. Neural Stem Cells
- 10.2.7. Others
- 11. Global Cell Therapy Market Analysis, by Application
- 11.1. Key Segment Analysis
- 11.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Application, 2021-2035
- 11.2.1. Oncology
- 11.2.2. Cardiovascular Diseases
- 11.2.3. Neurological Disorders
- 11.2.4. Musculoskeletal Disorders
- 11.2.5. Dermatology
- 11.2.6. Ophthalmology
- 11.2.7. Autoimmune Diseases
- 11.2.8. Wound Healing and Tissue Repair
- 11.2.9. Others
- 12. Global Cell Therapy Market Analysis, by End User
- 12.1. Key Segment Analysis
- 12.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by End User, 2021-2035
- 12.2.1. Hospitals and Clinics
- 12.2.2. Biopharmaceutical and Biotechnology Companies
- 12.2.3. Research Institutes
- 12.2.4. Contract Research Organizations (CROs)
- 12.2.5. Regenerative Medicine Centers
- 12.2.6. Others
- 13. Global Cell Therapy Market Analysis and Forecasts, by Region
- 13.1. Key Findings
- 13.2. Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 13.2.1. North America
- 13.2.2. Europe
- 13.2.3. Asia Pacific
- 13.2.4. Middle East
- 13.2.5. Africa
- 13.2.6. South America
- 14. North America Cell Therapy Market Analysis
- 14.1. Key Segment Analysis
- 14.2. Regional Snapshot
- 14.3. North America Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 14.3.1. Therapy Type
- 14.3.2. Cell Type
- 14.3.3. Technology
- 14.3.4. Therapy Technique
- 14.3.5. Cell Source
- 14.3.6. Application
- 14.3.7. End User
- 14.3.8. Country
- 14.3.8.1. USA
- 14.3.8.2. Canada
- 14.3.8.3. Mexico
- 14.4. USA Cell Therapy Market
- 14.4.1. Country Segmental Analysis
- 14.4.2. Therapy Type
- 14.4.3. Cell Type
- 14.4.4. Technology
- 14.4.5. Therapy Technique
- 14.4.6. Cell Source
- 14.4.7. Application
- 14.4.8. End User
- 14.5. Canada Cell Therapy Market
- 14.5.1. Country Segmental Analysis
- 14.5.2. Therapy Type
- 14.5.3. Cell Type
- 14.5.4. Technology
- 14.5.5. Therapy Technique
- 14.5.6. Cell Source
- 14.5.7. Application
- 14.5.8. End User
- 14.6. Mexico Cell Therapy Market
- 14.6.1. Country Segmental Analysis
- 14.6.2. Therapy Type
- 14.6.3. Cell Type
- 14.6.4. Technology
- 14.6.5. Therapy Technique
- 14.6.6. Cell Source
- 14.6.7. Application
- 14.6.8. End User
- 15. Europe Cell Therapy Market Analysis
- 15.1. Key Segment Analysis
- 15.2. Regional Snapshot
- 15.3. Europe Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 15.3.1. Therapy Type
- 15.3.2. Cell Type
- 15.3.3. Technology
- 15.3.4. Therapy Technique
- 15.3.5. Cell Source
- 15.3.6. Application
- 15.3.7. End User
- 15.3.8. Country
- 15.3.8.1. Germany
- 15.3.8.2. United Kingdom
- 15.3.8.3. France
- 15.3.8.4. Italy
- 15.3.8.5. Spain
- 15.3.8.6. Netherlands
- 15.3.8.7. Nordic Countries
- 15.3.8.8. Poland
- 15.3.8.9. Russia & CIS
- 15.3.8.10. Rest of Europe
- 15.4. Germany Cell Therapy Market
- 15.4.1. Country Segmental Analysis
- 15.4.2. Therapy Type
- 15.4.3. Cell Type
- 15.4.4. Technology
- 15.4.5. Therapy Technique
- 15.4.6. Cell Source
- 15.4.7. Application
- 15.4.8. End User
- 15.5. United Kingdom Cell Therapy Market
- 15.5.1. Country Segmental Analysis
- 15.5.2. Therapy Type
- 15.5.3. Cell Type
- 15.5.4. Technology
- 15.5.5. Therapy Technique
- 15.5.6. Cell Source
- 15.5.7. Application
- 15.5.8. End User
- 15.6. France Cell Therapy Market
- 15.6.1. Country Segmental Analysis
- 15.6.2. Therapy Type
- 15.6.3. Cell Type
- 15.6.4. Technology
- 15.6.5. Therapy Technique
- 15.6.6. Cell Source
- 15.6.7. Application
- 15.6.8. End User
- 15.7. Italy Cell Therapy Market
- 15.7.1. Country Segmental Analysis
- 15.7.2. Therapy Type
- 15.7.3. Cell Type
- 15.7.4. Technology
- 15.7.5. Therapy Technique
- 15.7.6. Cell Source
- 15.7.7. Application
- 15.7.8. End User
- 15.8. Spain Cell Therapy Market
- 15.8.1. Country Segmental Analysis
- 15.8.2. Therapy Type
- 15.8.3. Cell Type
- 15.8.4. Technology
- 15.8.5. Therapy Technique
- 15.8.6. Cell Source
- 15.8.7. Application
- 15.8.8. End User
- 15.9. Netherlands Cell Therapy Market
- 15.9.1. Country Segmental Analysis
- 15.9.2. Therapy Type
- 15.9.3. Cell Type
- 15.9.4. Technology
- 15.9.5. Therapy Technique
- 15.9.6. Cell Source
- 15.9.7. Application
- 15.9.8. End User
- 15.10. Nordic Countries Cell Therapy Market
- 15.10.1. Country Segmental Analysis
- 15.10.2. Therapy Type
- 15.10.3. Cell Type
- 15.10.4. Technology
- 15.10.5. Therapy Technique
- 15.10.6. Cell Source
- 15.10.7. Application
- 15.10.8. End User
- 15.11. Poland Cell Therapy Market
- 15.11.1. Country Segmental Analysis
- 15.11.2. Therapy Type
- 15.11.3. Cell Type
- 15.11.4. Technology
- 15.11.5. Therapy Technique
- 15.11.6. Cell Source
- 15.11.7. Application
- 15.11.8. End User
- 15.12. Russia & CIS Cell Therapy Market
- 15.12.1. Country Segmental Analysis
- 15.12.2. Therapy Type
- 15.12.3. Cell Type
- 15.12.4. Technology
- 15.12.5. Therapy Technique
- 15.12.6. Cell Source
- 15.12.7. Application
- 15.12.8. End User
- 15.13. Rest of Europe Cell Therapy Market
- 15.13.1. Country Segmental Analysis
- 15.13.2. Therapy Type
- 15.13.3. Cell Type
- 15.13.4. Technology
- 15.13.5. Therapy Technique
- 15.13.6. Cell Source
- 15.13.7. Application
- 15.13.8. End User
- 16. Asia Pacific Cell Therapy Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. Asia Pacific Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Therapy Type
- 16.3.2. Cell Type
- 16.3.3. Technology
- 16.3.4. Therapy Technique
- 16.3.5. Cell Source
- 16.3.6. Application
- 16.3.7. End User
- 16.3.8. Country
- 16.3.8.1. China
- 16.3.8.2. India
- 16.3.8.3. Japan
- 16.3.8.4. South Korea
- 16.3.8.5. Australia and New Zealand
- 16.3.8.6. Indonesia
- 16.3.8.7. Malaysia
- 16.3.8.8. Thailand
- 16.3.8.9. Vietnam
- 16.3.8.10. Rest of Asia Pacific
- 16.4. China Cell Therapy Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Therapy Type
- 16.4.3. Cell Type
- 16.4.4. Technology
- 16.4.5. Therapy Technique
- 16.4.6. Cell Source
- 16.4.7. Application
- 16.4.8. End User
- 16.5. India Cell Therapy Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Therapy Type
- 16.5.3. Cell Type
- 16.5.4. Technology
- 16.5.5. Therapy Technique
- 16.5.6. Cell Source
- 16.5.7. Application
- 16.5.8. End User
- 16.6. Japan Cell Therapy Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Therapy Type
- 16.6.3. Cell Type
- 16.6.4. Technology
- 16.6.5. Therapy Technique
- 16.6.6. Cell Source
- 16.6.7. Application
- 16.6.8. End User
- 16.7. South Korea Cell Therapy Market
- 16.7.1. Country Segmental Analysis
- 16.7.2. Therapy Type
- 16.7.3. Cell Type
- 16.7.4. Technology
- 16.7.5. Therapy Technique
- 16.7.6. Cell Source
- 16.7.7. Application
- 16.7.8. End User
- 16.8. Australia and New Zealand Cell Therapy Market
- 16.8.1. Country Segmental Analysis
- 16.8.2. Therapy Type
- 16.8.3. Cell Type
- 16.8.4. Technology
- 16.8.5. Therapy Technique
- 16.8.6. Cell Source
- 16.8.7. Application
- 16.8.8. End User
- 16.9. Indonesia Cell Therapy Market
- 16.9.1. Country Segmental Analysis
- 16.9.2. Therapy Type
- 16.9.3. Cell Type
- 16.9.4. Technology
- 16.9.5. Therapy Technique
- 16.9.6. Cell Source
- 16.9.7. Application
- 16.9.8. End User
- 16.10. Malaysia Cell Therapy Market
- 16.10.1. Country Segmental Analysis
- 16.10.2. Therapy Type
- 16.10.3. Cell Type
- 16.10.4. Technology
- 16.10.5. Therapy Technique
- 16.10.6. Cell Source
- 16.10.7. Application
- 16.10.8. End User
- 16.11. Thailand Cell Therapy Market
- 16.11.1. Country Segmental Analysis
- 16.11.2. Therapy Type
- 16.11.3. Cell Type
- 16.11.4. Technology
- 16.11.5. Therapy Technique
- 16.11.6. Cell Source
- 16.11.7. Application
- 16.11.8. End User
- 16.12. Vietnam Cell Therapy Market
- 16.12.1. Country Segmental Analysis
- 16.12.2. Therapy Type
- 16.12.3. Cell Type
- 16.12.4. Technology
- 16.12.5. Therapy Technique
- 16.12.6. Cell Source
- 16.12.7. Application
- 16.12.8. End User
- 16.13. Rest of Asia Pacific Cell Therapy Market
- 16.13.1. Country Segmental Analysis
- 16.13.2. Therapy Type
- 16.13.3. Cell Type
- 16.13.4. Technology
- 16.13.5. Therapy Technique
- 16.13.6. Cell Source
- 16.13.7. Application
- 16.13.8. End User
- 17. Middle East Cell Therapy Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Middle East Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Therapy Type
- 17.3.2. Cell Type
- 17.3.3. Technology
- 17.3.4. Therapy Technique
- 17.3.5. Cell Source
- 17.3.6. Application
- 17.3.7. End User
- 17.3.8. Country
- 17.3.8.1. Turkey
- 17.3.8.2. UAE
- 17.3.8.3. Saudi Arabia
- 17.3.8.4. Israel
- 17.3.8.5. Rest of Middle East
- 17.4. Turkey Cell Therapy Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Therapy Type
- 17.4.3. Cell Type
- 17.4.4. Technology
- 17.4.5. Therapy Technique
- 17.4.6. Cell Source
- 17.4.7. Application
- 17.4.8. End User
- 17.5. UAE Cell Therapy Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Therapy Type
- 17.5.3. Cell Type
- 17.5.4. Technology
- 17.5.5. Therapy Technique
- 17.5.6. Cell Source
- 17.5.7. Application
- 17.5.8. End User
- 17.6. Saudi Arabia Cell Therapy Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Therapy Type
- 17.6.3. Cell Type
- 17.6.4. Technology
- 17.6.5. Therapy Technique
- 17.6.6. Cell Source
- 17.6.7. Application
- 17.6.8. End User
- 17.7. Israel Cell Therapy Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Therapy Type
- 17.7.3. Cell Type
- 17.7.4. Technology
- 17.7.5. Therapy Technique
- 17.7.6. Cell Source
- 17.7.7. Application
- 17.7.8. End User
- 17.8. Rest of Middle East Cell Therapy Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Therapy Type
- 17.8.3. Cell Type
- 17.8.4. Technology
- 17.8.5. Therapy Technique
- 17.8.6. Cell Source
- 17.8.7. Application
- 17.8.8. End User
- 18. Africa Cell Therapy Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Africa Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Therapy Type
- 18.3.2. Cell Type
- 18.3.3. Technology
- 18.3.4. Therapy Technique
- 18.3.5. Cell Source
- 18.3.6. Application
- 18.3.7. End User
- 18.3.8. Country
- 18.3.8.1. South Africa
- 18.3.8.2. Egypt
- 18.3.8.3. Nigeria
- 18.3.8.4. Algeria
- 18.3.8.5. Rest of Africa
- 18.4. South Africa Cell Therapy Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Therapy Type
- 18.4.3. Cell Type
- 18.4.4. Technology
- 18.4.5. Therapy Technique
- 18.4.6. Cell Source
- 18.4.7. Application
- 18.4.8. End User
- 18.5. Egypt Cell Therapy Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Therapy Type
- 18.5.3. Cell Type
- 18.5.4. Technology
- 18.5.5. Therapy Technique
- 18.5.6. Cell Source
- 18.5.7. Application
- 18.5.8. End User
- 18.6. Nigeria Cell Therapy Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Therapy Type
- 18.6.3. Cell Type
- 18.6.4. Technology
- 18.6.5. Therapy Technique
- 18.6.6. Cell Source
- 18.6.7. Application
- 18.6.8. End User
- 18.7. Algeria Cell Therapy Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Therapy Type
- 18.7.3. Cell Type
- 18.7.4. Technology
- 18.7.5. Therapy Technique
- 18.7.6. Cell Source
- 18.7.7. Application
- 18.7.8. End User
- 18.8. Rest of Africa Cell Therapy Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Therapy Type
- 18.8.3. Cell Type
- 18.8.4. Technology
- 18.8.5. Therapy Technique
- 18.8.6. Cell Source
- 18.8.7. Application
- 18.8.8. End User
- 19. South America Cell Therapy Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. South America Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Therapy Type
- 19.3.2. Cell Type
- 19.3.3. Technology
- 19.3.4. Therapy Technique
- 19.3.5. Cell Source
- 19.3.6. Application
- 19.3.7. End User
- 19.3.8. Country
- 19.3.8.1. Brazil
- 19.3.8.2. Argentina
- 19.3.8.3. Rest of South America
- 19.4. Brazil Cell Therapy Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Therapy Type
- 19.4.3. Cell Type
- 19.4.4. Technology
- 19.4.5. Therapy Technique
- 19.4.6. Cell Source
- 19.4.7. Application
- 19.4.8. End User l
- 19.5. Argentina Cell Therapy Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Therapy Type
- 19.5.3. Cell Type
- 19.5.4. Technology
- 19.5.5. Therapy Technique
- 19.5.6. Cell Source
- 19.5.7. Application
- 19.5.8. End User
- 19.6. Rest of South America Cell Therapy Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Therapy Type
- 19.6.3. Cell Type
- 19.6.4. Technology
- 19.6.5. Therapy Technique
- 19.6.6. Cell Source
- 19.6.7. Application
- 19.6.8. End User
- 20. Key Players/ Company Profile
- 20.1. Allogene Therapeutics, Inc.
- 20.1.1. Company Details/ Overview
- 20.1.2. Company Financials
- 20.1.3. Key Customers and Competitors
- 20.1.4. Business/ Industry Portfolio
- 20.1.5. Product Portfolio/ Specification Details
- 20.1.6. Pricing Data
- 20.1.7. Strategic Overview
- 20.1.8. Recent Developments
- 20.2. Amgen Inc.
- 20.3. Astellas Pharma Inc.
- 20.4. Atara Biotherapeutics, Inc.
- 20.5. Autolus Therapeutics plc
- 20.6. bluebird bio, Inc.
- 20.7. Bristol-Myers Squibb Company
- 20.8. CARsgen Therapeutics Holdings Limited
- 20.9. Cellectis S.A.
- 20.10. CRISPR Therapeutics AG
- 20.11. Fate Therapeutics, Inc.
- 20.12. Gilead Sciences, Inc.
- 20.13. Iovance Biotherapeutics, Inc.
- 20.14. Johnson & Johnson
- 20.15. JW Therapeutics (Shanghai) Co., Ltd.
- 20.16. Legend Biotech Corporation
- 20.17. Mesoblast Limited
- 20.18. Novartis AG
- 20.19. Sangamo Therapeutics, Inc.
- 20.20. Vericel Corporation
- 20.21. Other Key Players
- 20.1. Allogene Therapeutics, Inc.
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
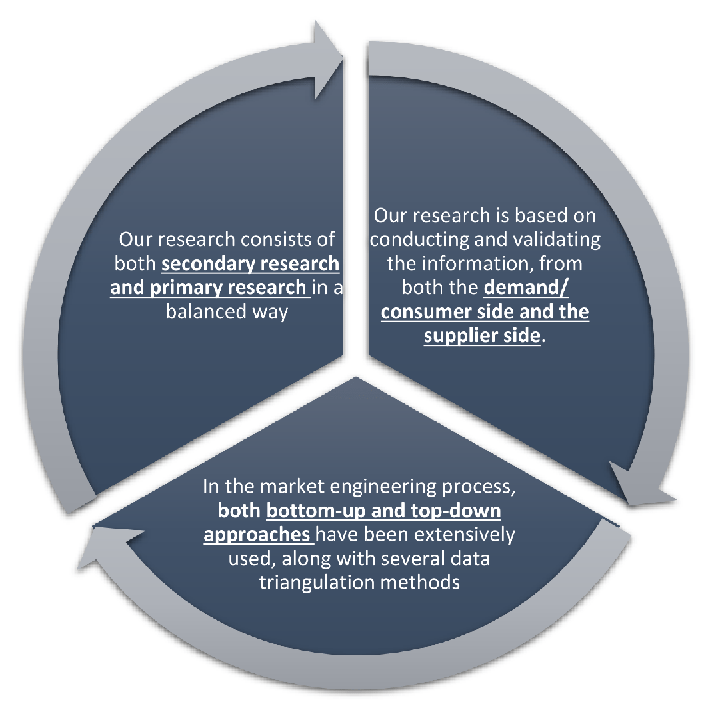
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
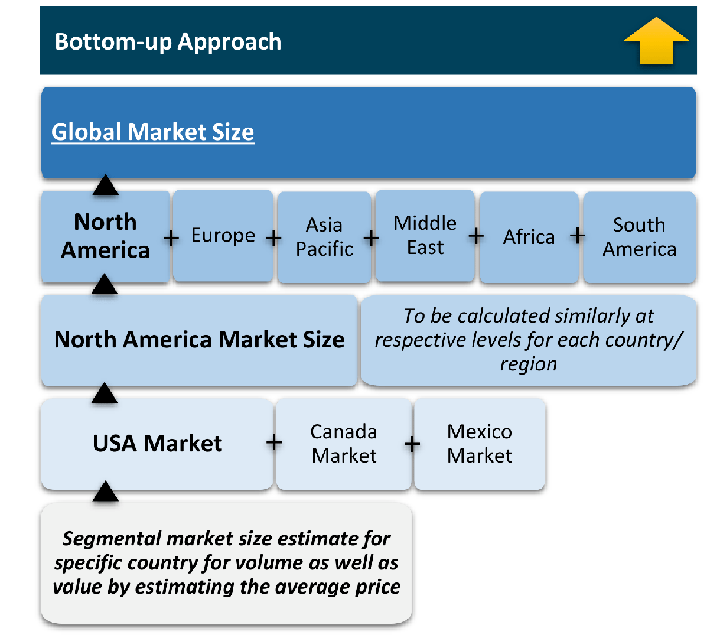
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
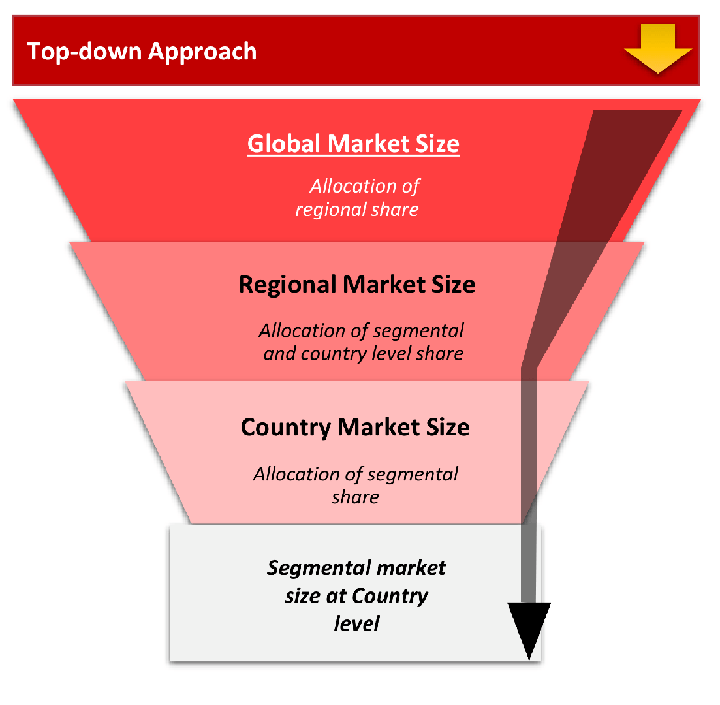
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
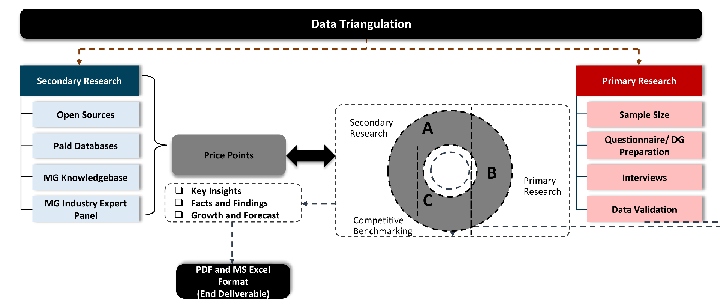
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation