Cryoablation Devices Market Size, Share & Trends Analysis Report by Product Type (Cryoablation Catheters, Cryoablation Probes, Cryoablation Consoles/Systems, Cryosurgical Units, Accessories and Consumables), Technology, Therapy Type, Application, End-Users, Imaging Guidance Distribution Channel, and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Cryoablation Devices Market Size, Share, and Growth
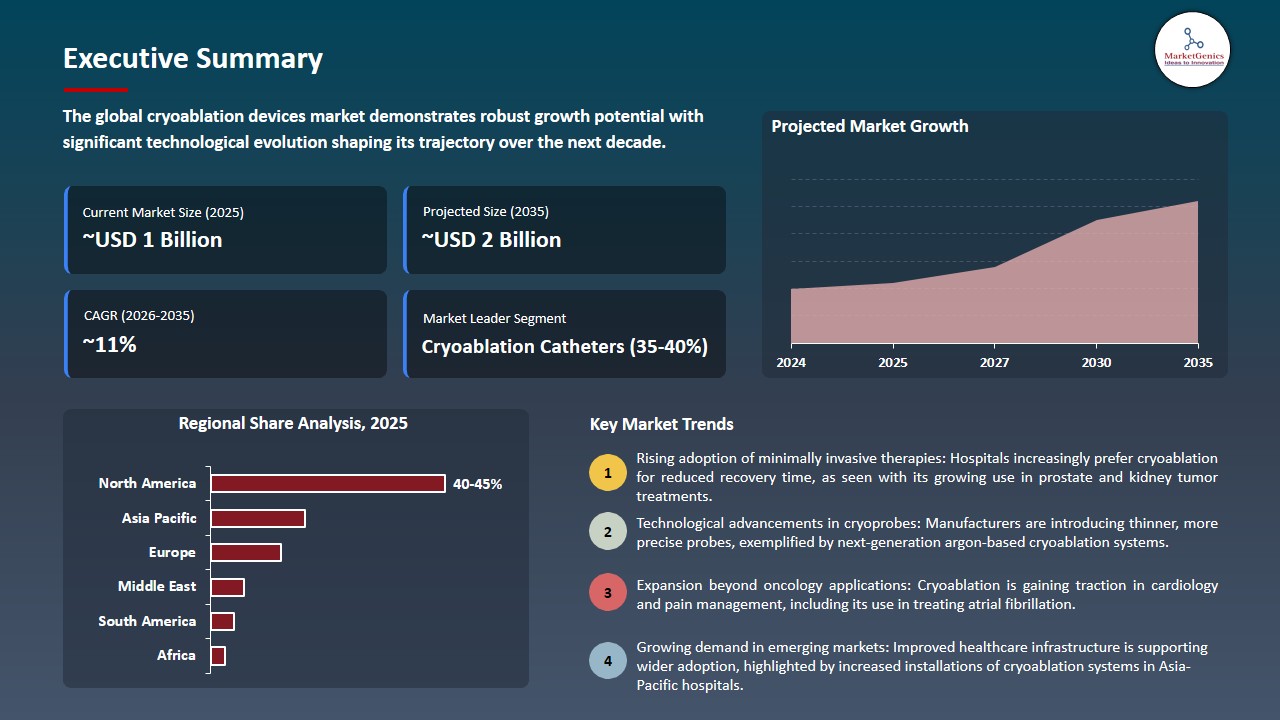
The global cryoablation devices market is witnessing strong growth, valued at USD 0.7 billion in 2025 and projected to reach ~USD 2 billion by 2035, expanding at a CAGR of 11.2% during the forecast period. Asia Pacific is the fastest-growing cryoablation devices market due to rapidly expanding healthcare infrastructure, rising prevalence of cardiovascular diseases and cancer, increasing adoption of minimally invasive procedures, and improving access to advanced medical technologies across emerging economies.
Eyal Shamir, Chief Executive Officer, IceCure, said, “We are excited to add a minimally invasive choice around breast cancer treatments and to offer patients an effective, outpatient procedure, With the ProSense Cryoablation System, we are giving women with low-risk, early-stage breast cancer the choice to freeze their cancer, not their lives, through an effective treatment that minimizes recovery time, and minimal cosmetic changes to the breast”.
The increased prevalence of solid tumors, especially liver, kidney, and lung cancers is fueling the use of cryoablation therapies in the world. Cryoablation provides the patient that cannot undergo surgery with a minimally invasive, targeted and organ-sparing treatment that cuts down on recovery time and complications. This trend amongst clinical preferences, coupled with a rising incidence of cancer diagnosis, is creating a great deal of demand in the purchase of sophisticated cryoablation equipment across hospitals, outpatient care facilities, and oncology centers.
The innovation of the portable and outpatient cryoablation systems offers a great growth potential to the cryoablation devices market. The miniature and easy to use machines allow minimum invasive surgery beyond the walls of a hospital, such as outpatient clinics and ambulatory care centers, to decrease the cost of treatment and the recovery period of patients. The general penetration of cryoablation therapies across the globe can be enhanced by the wider adoption of such systems in emerging markets, procedural volumes, and the accessibility of such systems.
Key adjacent opportunities for the cryoablation devices market include cardiac rhythm management devices, renal and hepatic tumor ablation technologies, minimally invasive surgical tools, image-guided intervention systems, and oncology-focused energy-based ablation devices. Expanding into these areas can drive cross-market adoption and technological integration, potentially increasing market growth and revenue diversification.

Cryoablation Devices Market Dynamics and Trends
Driver: Increasing Clinical Adoption Due to Expanded Regulatory Authorizations in Oncology Treatment Settings
-
Extended regulatory licenses are playing a major role in hastening clinical incorporation of the cryoablation devices in the cancer care by officially approving their safety and efficacy and therapeutic significance. The regulatory approvals would allow clinicians to be more comfortable to incorporate cryoablation into the conventional treatment protocols, especially in the management of low-risk and early cancer where less invasive procedures are becoming a more popular choice.
- Recent approvals have also enhanced the transparency of reimbursement and promoted the adoption of cryoablation platforms by outpatient and day-care oncology facilities by hospital systems. With regulatory bodies recognizing the use of cryoablation as an alternative to surgical intervention, manufacturers will experience a wider market access, and physicians will suffer more flexibility in the procedures and an accepted procedure by patients.
- IceCure Medical was the first and only company to be granted U.S. FDA marketing approval for its ProSense Cryoablation System in October 2025, becoming the first and only device authorized for local treatment of early minor risk breast cancer in women aged 70+, increasing treatment options in patients who would otherwise have an estimated 46,000 annually, which contributes to the credibility of the clinical use of cryoablation in the treatment of cancer.
- This regulatory impetus directly contributes to the increase in proceedings volumes, increased indications, and the long-term increase in the market of cryoablation devices.
Restraint: High Capital Investment Burdens Adoption Across Healthcare Facility Types
-
High capital cost of the device is a huge limitation to the cryoablation devices market because the technology is intensive in upfront spending on the consoles, cryogen supply system, advanced imaging compatibility and specialized consumables. Cryoablation systems at high unit costs with recurrent costs on probes and maintenance cause financial strain to healthcare providers especially when compared to more proven thermal ablation options which have lower entry costs.
- Adoption is also limited among the various types of healthcare facilities as a result of disparity in the availability of capital. Large tertiary hospitals can cover the investment by high patient volumes and multi-specialty use, whereas mid-sized hospitals, specialty clinics, and ambulatory surgical centers have budget constraints that postpone the decision-making process of purchasing.
- The lack of resources in healthcare systems and effective reimbursement systems in the developing world only exacerbates this obstacle and restricts the broader spread of cryoablation technology, despite continued growth in clinical demand.
Opportunity: Expanding Application Scope Beyond Traditional Cardiac and Tumor Ablation Indications
-
The cryoablation devices market has a strong growth potential of expansion into non-traditional clinical uses besides cardiac arrhythmia and solid tumor ablation. The diversity and accuracy of the technology in clinical applications are demonstrated by its increasing use in chronic pain management, gynecology, urology, dermatology and musculoskeletal disorders.
- Cryoablation has the ability to spare tissue, lower the pain during the procedure and recover quickly, which is why it is appropriate in nerve ablation, benign lesion and outpatient surgery procedures. Improvement of probe design and imaging guidance are additional pushes towards safe adoption of the same across specialties.
- In August 2023, Boston Scientific was granted FDA 510(k) clearance to an expanded indication of its Visual ICE Cryoablation System to address pain in patients with bone metastases who do not respond to or cannot receive radiation therapy.
- The clinical expansion of this system to palliative pain was made possible by this clearance, extending the clinical application of cryoablation into the sphere of palliative pain management and displaying the versatility of the technique in palliative pain management, ablation of solid tumors, and quality-of-life enhancement applications.
Key Trend: Integration of Real-Time Imaging and Navigation Software Enhances Procedural Precision
-
A major development in the market of cryoablation devices is the incorporation of real-time imaging and navigation software to enhance the accuracy and outcomes of the procedures. The highly developed imaging techniques like MRI, CT and high-resolution ultrasound enable clinicians to observe iceball development and tissue contact during ablation, and the precise targeting is achieved without inflicting severe injury on the surrounding normal structures.
- The intelligent navigation software with cryoablation platform lessens dependency on operators, decreases learning curves, and increases the safety of the procedures. Complex and multi-site ablations are also backed up by this trend, and more controlled conditions are treated with high precision.
- In October 2025, Merit medical Systems bought the C2 CryoBalloon technology of PENTAX medical which is a minimally invasive technology aimed at treating Barretts Esophagus and other gastrointestinal diseases.
- The device is designed to provide controlled cryotherapy and maintain the surrounding tissues, and with Merit managing it, it is anticipated to increase its adoption and become part of their endoscopy and interventional imaging solutions, and aid real-time, precision-guided ablation procedures and improve clinical outcomes in minimally invasive GI treatments.
- Technological convergence enhances clinician confidence, adoption, and distinguishes manufacturers in a competitive market.
Cryoablation-Devices-Market Analysis and Segmental Data
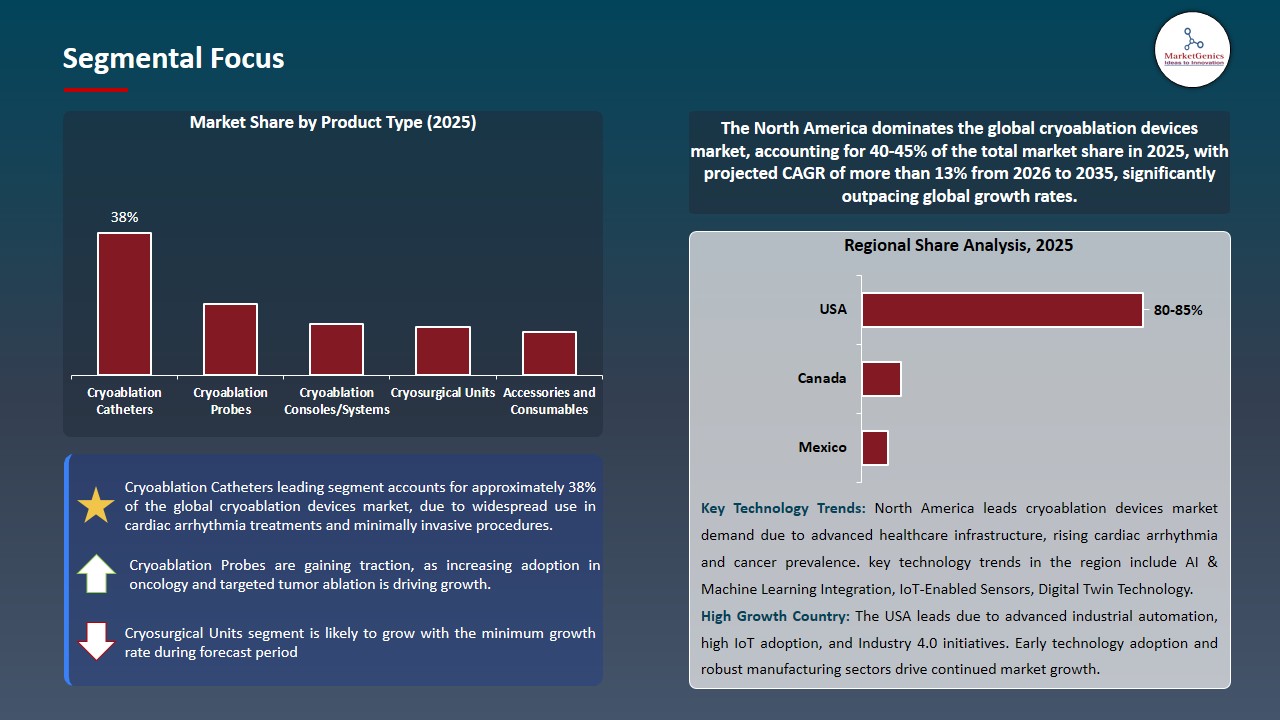
Cryoablation Catheters Dominate Global Cryoablation Devices Market
-
Cryoablation catheters remain the largest and most widely adopted segment within the global cryoablation devices market due to their established efficacy in treating cardiac arrhythmias, particularly atrial fibrillation, and solid tumors. Their minimally invasive design, combined with precision-controlled cryotherapy, allows for targeted ablation of diseased tissue while preserving surrounding structures, reducing procedural complications and recovery times.
- This has been achieved through continuous innovation in catheter design such as greater flexibility, steerable, and integrated imaging guidance that has increased the success of the procedure and confidence of the clinician. Also, the dominance is supported by the fact that they are highly available, have a strong clinical evidence, and have regulatory approvals in various geographies.
- Shanghai Antec Medical Technology Co., Ltd. was authorized in February 2025, through a Cryoablation Console with a 32 mm balloon cryoablation catheter, to treat recurrent, symptomatic paroxysmal atrial fibrillation.
- The uniform usage of the segment in the hospitals, heart centers, and outpatient facilities guarantee the market leadership and promote the total revenue growth of the cryoablation equipment vendors.
North America Leads Global Cryoablation Devices Market Demand
-
North America continues to dominate the global cryoablation devices market due to a combination of advanced healthcare infrastructure, high adoption of minimally invasive procedures, and favorable reimbursement policies. The region benefits from a well-established network of hospitals, specialty cardiac centers, and oncology clinics that actively invest in cutting-edge technologies, supporting widespread clinical adoption of cryoablation devices for cardiac arrhythmias, solid tumors, and emerging therapeutic indications.
- The availability of major players in the market with high level of R&D and big product lines contribute to the regional demand even more. Also, continued regulatory endorsement, awareness campaigns and clinical evidence that cryoablation is effective and safe motivate clinicians and patients to use cryoablation instead of traditional methods of surgery.
- High procedure volumes, early uptake of next-generation devices incorporating imaging and navigation technologies make North America the largest and most profitable regional market which sets trends that determine development and adoption trends of cryoablation devices worldwide.
Cryoablation-Devices-Market Ecosystem
The global cryoablation devices market is consolidated, with key players including Medtronic, Boston Scientific Corporation, AtriCure, IceCure Medical, and Adagio Medical Inc. These companies maintain competitive positions through robust research and development capabilities, innovation in catheter and console technologies, and expertise in minimally invasive ablation procedures. Their leadership is reinforced by long-standing relationships with hospitals, cardiac and oncology centers, and outpatient clinics, as well as global distribution networks and strict compliance with regulatory standards, including FDA, CE, and ISO certifications.
The market value chain encompasses the design and development of cryoablation consoles and catheters, integration with real-time imaging and navigation software, device customization for specific clinical applications, on-site installation and training, and post-deployment support including maintenance and procedural guidance. These stages ensure procedural efficacy, patient safety, and regulatory compliance while supporting smooth clinical adoption.
High entry barriers exist due to substantial capital investment, advanced technological expertise, and stringent regulatory requirements. Continuous innovations such as integrated imaging systems, balloon and catheter design improvements, and AI-assisted procedural planning drive product differentiation, enhance procedural accuracy, and support sustained global market growth.

Recent Development and Strategic Overview:
-
In April 2024, AtriCure launched the cryoSPHERE+ cryoablation probe, featuring new insulation technology that reduces freeze times by 25% compared to the legacy cryoSPHERE device. The FDA-cleared device temporarily blocks pain by ablating peripheral and intercostal nerves in adults and adolescents, enabling faster procedures and improved patient outcomes.
- In April 2024, IceCure Medical, a innovator in minimally invasive therapies, received FDA 510(k) clearance for its XSense Cryoablation System with CryoProbes. This regulatory milestone enables U.S. commercialization, positioning the company to expand its footprint in the growing cryoablation devices market.
Report Scope
|
Detail |
|
|
Market Size in 2025 |
USD 0.7 Bn |
|
Market Forecast Value in 2035 |
~USD 2 Bn |
|
Growth Rate (CAGR) |
11.2% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
US$ Billion for Value Thousand Units for Volume |
|
Report Format |
Electronic (PDF) + Excel |
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Cryoablation-Devices-Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Cryoablation Devices Market, By Product Type |
|
|
Cryoablation Devices Market, By Technology |
|
|
Cryoablation Devices Market, By Therapy Type |
|
|
Cryoablation Devices Market, By Application |
|
|
Cryoablation Devices Market, By End-Users |
|
|
Cryoablation Devices Market, By Imaging Guidance |
|
|
Cryoablation Devices Market, By Distribution Channel |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Cryoablation Devices Market Outlook
- 2.1.1. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Cryoablation Devices Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Industry Ecosystem Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.4.1. Import & Export Analysis, 2025
- 3.4.2. Top Importing Countries
- 3.4.3. Top Exporting Countries
- 3.5. Trump Tariff Impact Analysis
- 3.5.1. Manufacturer
- 3.5.1.1. Based on the component & Raw material
- 3.5.2. Supply Chain
- 3.5.3. End Consumer
- 3.5.1. Manufacturer
- 3.6. Raw Material Analysis
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Increasing incidence of cancer and cardiac arrhythmias driving demand for cryoablation procedures.
- 4.1.1.2. Growing adoption of minimally invasive treatments due to reduced recovery time and complications.
- 4.1.1.3. Continuous technological advancements improving precision and clinical outcomes.
- 4.1.2. Restraints
- 4.1.2.1. High cost of cryoablation devices and procedures.
- 4.1.2.2. Limited availability of skilled professionals and infrastructure in emerging regions.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.4.1. Component Suppliers
- 4.4.2. Cryoablation Device Manufacturers
- 4.4.3. System Integrators
- 4.4.4. Distributors & Medical Device Logistics Providers
- 4.4.5. Healthcare Provider Adoption
- 4.4.6. End Users
- 4.5. Cost Structure Analysis
- 4.5.1. Parameter’s Share for Cost Associated
- 4.5.2. COGP vs COGS
- 4.5.3. Profit Margin Analysis
- 4.6. Pricing Analysis
- 4.6.1. Regional Pricing Analysis
- 4.6.2. Segmental Pricing Trends
- 4.6.3. Factors Influencing Pricing
- 4.7. Porter’s Five Forces Analysis
- 4.8. PESTEL Analysis
- 4.9. Global Cryoablation Devices Market Demand
- 4.9.1. Historical Market Size – Volume (Thousand Units) and Value (US$ Bn), 2020-2024
- 4.9.2. Current and Future Market Size – Volume (Thousand Units) and Value (US$ Bn), 2026–2035
- 4.9.2.1. Y-o-Y Growth Trends
- 4.9.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Cryoablation Devices Market Analysis, by Product Type
- 6.1. Key Segment Analysis
- 6.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Product Type, 2021-2035
- 6.2.1. Cryoablation Catheters
- 6.2.1.1. Focal Cryoablation Catheters
- 6.2.1.2. Linear Cryoablation Catheters
- 6.2.2. Cryoablation Probes
- 6.2.2.1. Single-Use Probes
- 6.2.2.2. Reusable Probes
- 6.2.3. Cryoablation Consoles/Systems
- 6.2.4. Cryosurgical Units
- 6.2.5. Accessories and Consumables
- 6.2.1. Cryoablation Catheters
- 7. Global Cryoablation Devices Market Analysis, by Technology
- 7.1. Key Segment Analysis
- 7.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Technology, 2021-2035
- 7.2.1. Liquid Nitrogen-Based Cryoablation
- 7.2.2. Argon Gas-Based Cryoablation
- 7.2.3. Nitrous Oxide-Based Cryoablation
- 7.2.4. Carbon Dioxide-Based Cryoablation
- 8. Global Cryoablation Devices Market Analysis, by Therapy Type
- 8.1. Key Segment Analysis
- 8.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Therapy Type, 2021-2035
- 8.2.1. Cryosurgery
- 8.2.2. Cryotherapy
- 8.2.3. Image-Guided Cryoablation
- 8.2.4. Percutaneous Cryoablation
- 9. Global Cryoablation Devices Market Analysis, by Application
- 9.1. Key Segment Analysis
- 9.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Application, 2021-2035
- 9.2.1. Cardiac Arrhythmia
- 9.2.1.1. Atrial Fibrillation
- 9.2.1.2. Atrial Flutter
- 9.2.1.3. Ventricular Tachycardia
- 9.2.1.4. Supraventricular Tachycardia
- 9.2.1.5. Others
- 9.2.2. Oncology/Cancer Treatment
- 9.2.2.1. Prostate Cancer
- 9.2.2.2. Liver Cancer
- 9.2.2.3. Kidney Cancer
- 9.2.2.4. Lung Cancer
- 9.2.2.5. Breast Cancer
- 9.2.2.6. Bone Cancer
- 9.2.2.7. Skin Cancer
- 9.2.2.8. Others
- 9.2.3. Pain Management
- 9.2.3.1. Chronic Pain
- 9.2.3.2. Neuropathic Pain
- 9.2.3.3. Musculoskeletal Pain
- 9.2.3.4. Others
- 9.2.3.5. Dermatology
- 9.2.3.6. Skin Lesions
- 9.2.3.7. Warts
- 9.2.3.8. Benign Tumors
- 9.2.3.9. Others
- 9.2.4. Gynecology
- 9.2.4.1. Cervical Dysplasia
- 9.2.4.2. Uterine Fibroids
- 9.2.5. Others
- 9.2.1. Cardiac Arrhythmia
- 10. Global Cryoablation Devices Market Analysis, by End-Users
- 10.1. Key Segment Analysis
- 10.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by End-Users, 2021-2035
- 10.2.1. Hospitals
- 10.2.1.1. Multi-Specialty Hospitals
- 10.2.1.2. Specialty Hospitals
- 10.2.2. Ambulatory Surgical Centers
- 10.2.3. Cardiac Catheterization Laboratories
- 10.2.4. Cancer Treatment Centers/Oncology Clinics
- 10.2.5. Dermatology Clinics
- 10.2.6. Pain Management Centers
- 10.2.7. Academic and Research Institutes
- 10.2.8. Others
- 10.2.1. Hospitals
- 11. Global Cryoablation Devices Market Analysis, by Imaging Guidance
- 11.1. Key Segment Analysis
- 11.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Imaging Guidance, 2021-2035
- 11.2.1. Ultrasound-guided Cryoablation
- 11.2.2. CT-guided Cryoablation
- 11.2.3. MRI-guided Cryoablation
- 11.2.4. Fluoroscopy-guided Cryoablation
- 11.2.5. Non-guided Cryoablation
- 12. Global Cryoablation Devices Market Analysis, by Distribution Channel
- 12.1. Key Segment Analysis
- 12.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Distribution Channel, 2021-2035
- 12.2.1. Direct Sales
- 12.2.2. Distributors & Wholesalers
- 12.2.3. Online Platforms
- 13. Global Cryoablation Devices Market Analysis and Forecasts, by Region
- 13.1. Key Findings
- 13.2. Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 13.2.1. North America
- 13.2.2. Europe
- 13.2.3. Asia Pacific
- 13.2.4. Middle East
- 13.2.5. Africa
- 13.2.6. South America
- 14. North America Cryoablation Devices Market Analysis
- 14.1. Key Segment Analysis
- 14.2. Regional Snapshot
- 14.3. North America Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 14.3.1. Product Type
- 14.3.2. Technology
- 14.3.3. Therapy Type
- 14.3.4. Application
- 14.3.5. End-Users
- 14.3.6. Imaging Guidance
- 14.3.7. Distribution Channel
- 14.3.8. Country
- 14.3.8.1. USA
- 14.3.8.2. Canada
- 14.3.8.3. Mexico
- 14.4. USA Cryoablation Devices Market
- 14.4.1. Country Segmental Analysis
- 14.4.2. Product Type
- 14.4.3. Technology
- 14.4.4. Therapy Type
- 14.4.5. Application
- 14.4.6. End-Users
- 14.4.7. Imaging Guidance
- 14.4.8. Distribution Channel
- 14.5. Canada Cryoablation Devices Market
- 14.5.1. Country Segmental Analysis
- 14.5.2. Product Type
- 14.5.3. Technology
- 14.5.4. Therapy Type
- 14.5.5. Application
- 14.5.6. End-Users
- 14.5.7. Imaging Guidance
- 14.5.8. Distribution Channel
- 14.6. Mexico Cryoablation Devices Market
- 14.6.1. Country Segmental Analysis
- 14.6.2. Product Type
- 14.6.3. Technology
- 14.6.4. Therapy Type
- 14.6.5. Application
- 14.6.6. End-Users
- 14.6.7. Imaging Guidance
- 14.6.8. Distribution Channel
- 15. Europe Cryoablation Devices Market Analysis
- 15.1. Key Segment Analysis
- 15.2. Regional Snapshot
- 15.3. Europe Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 15.3.1. Product Type
- 15.3.2. Technology
- 15.3.3. Therapy Type
- 15.3.4. Application
- 15.3.5. End-Users
- 15.3.6. Imaging Guidance
- 15.3.7. Distribution Channel
- 15.3.8. Country
- 15.3.8.1. Germany
- 15.3.8.2. United Kingdom
- 15.3.8.3. France
- 15.3.8.4. Italy
- 15.3.8.5. Spain
- 15.3.8.6. Netherlands
- 15.3.8.7. Nordic Countries
- 15.3.8.8. Poland
- 15.3.8.9. Russia & CIS
- 15.3.8.10. Rest of Europe
- 15.4. Germany Cryoablation Devices Market
- 15.4.1. Country Segmental Analysis
- 15.4.2. Product Type
- 15.4.3. Technology
- 15.4.4. Therapy Type
- 15.4.5. Application
- 15.4.6. End-Users
- 15.4.7. Imaging Guidance
- 15.4.8. Distribution Channel
- 15.5. United Kingdom Cryoablation Devices Market
- 15.5.1. Country Segmental Analysis
- 15.5.2. Product Type
- 15.5.3. Technology
- 15.5.4. Therapy Type
- 15.5.5. Application
- 15.5.6. End-Users
- 15.5.7. Imaging Guidance
- 15.5.8. Distribution Channel
- 15.6. France Cryoablation Devices Market
- 15.6.1. Country Segmental Analysis
- 15.6.2. Product Type
- 15.6.3. Technology
- 15.6.4. Therapy Type
- 15.6.5. Application
- 15.6.6. End-Users
- 15.6.7. Imaging Guidance
- 15.6.8. Distribution Channel
- 15.7. Italy Cryoablation Devices Market
- 15.7.1. Country Segmental Analysis
- 15.7.2. Product Type
- 15.7.3. Technology
- 15.7.4. Therapy Type
- 15.7.5. Application
- 15.7.6. End-Users
- 15.7.7. Imaging Guidance
- 15.7.8. Distribution Channel
- 15.8. Spain Cryoablation Devices Market
- 15.8.1. Country Segmental Analysis
- 15.8.2. Product Type
- 15.8.3. Technology
- 15.8.4. Therapy Type
- 15.8.5. Application
- 15.8.6. End-Users
- 15.8.7. Imaging Guidance
- 15.8.8. Distribution Channel
- 15.9. Netherlands Cryoablation Devices Market
- 15.9.1. Country Segmental Analysis
- 15.9.2. Product Type
- 15.9.3. Technology
- 15.9.4. Therapy Type
- 15.9.5. Application
- 15.9.6. End-Users
- 15.9.7. Imaging Guidance
- 15.9.8. Distribution Channel
- 15.10. Nordic Countries Cryoablation Devices Market
- 15.10.1. Country Segmental Analysis
- 15.10.2. Product Type
- 15.10.3. Technology
- 15.10.4. Therapy Type
- 15.10.5. Application
- 15.10.6. End-Users
- 15.10.7. Imaging Guidance
- 15.10.8. Distribution Channel
- 15.11. Poland Cryoablation Devices Market
- 15.11.1. Country Segmental Analysis
- 15.11.2. Product Type
- 15.11.3. Technology
- 15.11.4. Therapy Type
- 15.11.5. Application
- 15.11.6. End-Users
- 15.11.7. Imaging Guidance
- 15.11.8. Distribution Channel
- 15.12. Russia & CIS Cryoablation Devices Market
- 15.12.1. Country Segmental Analysis
- 15.12.2. Product Type
- 15.12.3. Technology
- 15.12.4. Therapy Type
- 15.12.5. Application
- 15.12.6. End-Users
- 15.12.7. Imaging Guidance
- 15.12.8. Distribution Channel
- 15.13. Rest of Europe Cryoablation Devices Market
- 15.13.1. Country Segmental Analysis
- 15.13.2. Product Type
- 15.13.3. Technology
- 15.13.4. Therapy Type
- 15.13.5. Application
- 15.13.6. End-Users
- 15.13.7. Imaging Guidance
- 15.13.8. Distribution Channel
- 16. Asia Pacific Cryoablation Devices Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. Asia Pacific Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Product Type
- 16.3.2. Technology
- 16.3.3. Therapy Type
- 16.3.4. Application
- 16.3.5. End-Users
- 16.3.6. Imaging Guidance
- 16.3.7. Distribution Channel
- 16.3.8. Country
- 16.3.8.1. China
- 16.3.8.2. India
- 16.3.8.3. Japan
- 16.3.8.4. South Korea
- 16.3.8.5. Australia and New Zealand
- 16.3.8.6. Indonesia
- 16.3.8.7. Malaysia
- 16.3.8.8. Thailand
- 16.3.8.9. Vietnam
- 16.3.8.10. Rest of Asia Pacific
- 16.4. China Cryoablation Devices Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Product Type
- 16.4.3. Technology
- 16.4.4. Therapy Type
- 16.4.5. Application
- 16.4.6. End-Users
- 16.4.7. Imaging Guidance
- 16.4.8. Distribution Channel
- 16.5. India Cryoablation Devices Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Product Type
- 16.5.3. Technology
- 16.5.4. Therapy Type
- 16.5.5. Application
- 16.5.6. End-Users
- 16.5.7. Imaging Guidance
- 16.5.8. Distribution Channel
- 16.6. Japan Cryoablation Devices Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Product Type
- 16.6.3. Technology
- 16.6.4. Therapy Type
- 16.6.5. Application
- 16.6.6. End-Users
- 16.6.7. Imaging Guidance
- 16.6.8. Distribution Channel
- 16.7. South Korea Cryoablation Devices Market
- 16.7.1. Country Segmental Analysis
- 16.7.2. Product Type
- 16.7.3. Technology
- 16.7.4. Therapy Type
- 16.7.5. Application
- 16.7.6. End-Users
- 16.7.7. Imaging Guidance
- 16.7.8. Distribution Channel
- 16.8. Australia and New Zealand Cryoablation Devices Market
- 16.8.1. Country Segmental Analysis
- 16.8.2. Product Type
- 16.8.3. Technology
- 16.8.4. Therapy Type
- 16.8.5. Application
- 16.8.6. End-Users
- 16.8.7. Imaging Guidance
- 16.8.8. Distribution Channel
- 16.9. Indonesia Cryoablation Devices Market
- 16.9.1. Country Segmental Analysis
- 16.9.2. Product Type
- 16.9.3. Technology
- 16.9.4. Therapy Type
- 16.9.5. Application
- 16.9.6. End-Users
- 16.9.7. Imaging Guidance
- 16.9.8. Distribution Channel
- 16.10. Malaysia Cryoablation Devices Market
- 16.10.1. Country Segmental Analysis
- 16.10.2. Product Type
- 16.10.3. Technology
- 16.10.4. Therapy Type
- 16.10.5. Application
- 16.10.6. End-Users
- 16.10.7. Imaging Guidance
- 16.10.8. Distribution Channel
- 16.11. Thailand Cryoablation Devices Market
- 16.11.1. Country Segmental Analysis
- 16.11.2. Product Type
- 16.11.3. Technology
- 16.11.4. Therapy Type
- 16.11.5. Application
- 16.11.6. End-Users
- 16.11.7. Imaging Guidance
- 16.11.8. Distribution Channel
- 16.12. Vietnam Cryoablation Devices Market
- 16.12.1. Country Segmental Analysis
- 16.12.2. Product Type
- 16.12.3. Technology
- 16.12.4. Therapy Type
- 16.12.5. Application
- 16.12.6. End-Users
- 16.12.7. Imaging Guidance
- 16.12.8. Distribution Channel
- 16.13. Rest of Asia Pacific Cryoablation Devices Market
- 16.13.1. Country Segmental Analysis
- 16.13.2. Product Type
- 16.13.3. Technology
- 16.13.4. Therapy Type
- 16.13.5. Application
- 16.13.6. End-Users
- 16.13.7. Imaging Guidance
- 16.13.8. Distribution Channel
- 17. Middle East Cryoablation Devices Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Middle East Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Product Type
- 17.3.2. Technology
- 17.3.3. Therapy Type
- 17.3.4. Application
- 17.3.5. End-Users
- 17.3.6. Imaging Guidance
- 17.3.7. Distribution Channel
- 17.3.8. Country
- 17.3.8.1. Turkey
- 17.3.8.2. UAE
- 17.3.8.3. Saudi Arabia
- 17.3.8.4. Israel
- 17.3.8.5. Rest of Middle East
- 17.4. Turkey Cryoablation Devices Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Product Type
- 17.4.3. Technology
- 17.4.4. Therapy Type
- 17.4.5. Application
- 17.4.6. End-Users
- 17.4.7. Imaging Guidance
- 17.4.8. Distribution Channel
- 17.5. UAE Cryoablation Devices Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Product Type
- 17.5.3. Technology
- 17.5.4. Therapy Type
- 17.5.5. Application
- 17.5.6. End-Users
- 17.5.7. Imaging Guidance
- 17.5.8. Distribution Channel
- 17.6. Saudi Arabia Cryoablation Devices Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Product Type
- 17.6.3. Technology
- 17.6.4. Therapy Type
- 17.6.5. Application
- 17.6.6. End-Users
- 17.6.7. Imaging Guidance
- 17.6.8. Distribution Channel
- 17.7. Israel Cryoablation Devices Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Product Type
- 17.7.3. Technology
- 17.7.4. Therapy Type
- 17.7.5. Application
- 17.7.6. End-Users
- 17.7.7. Imaging Guidance
- 17.7.8. Distribution Channel
- 17.8. Rest of Middle East Cryoablation Devices Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Product Type
- 17.8.3. Technology
- 17.8.4. Therapy Type
- 17.8.5. Application
- 17.8.6. End-Users
- 17.8.7. Imaging Guidance
- 17.8.8. Distribution Channel
- 18. Africa Cryoablation Devices Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Africa Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Product Type
- 18.3.2. Technology
- 18.3.3. Therapy Type
- 18.3.4. Application
- 18.3.5. End-Users
- 18.3.6. Imaging Guidance
- 18.3.7. Distribution Channel
- 18.3.8. Country
- 18.3.8.1. South Africa
- 18.3.8.2. Egypt
- 18.3.8.3. Nigeria
- 18.3.8.4. Algeria
- 18.3.8.5. Rest of Africa
- 18.4. South Africa Cryoablation Devices Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Product Type
- 18.4.3. Technology
- 18.4.4. Therapy Type
- 18.4.5. Application
- 18.4.6. End-Users
- 18.4.7. Imaging Guidance
- 18.4.8. Distribution Channel
- 18.5. Egypt Cryoablation Devices Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Product Type
- 18.5.3. Technology
- 18.5.4. Therapy Type
- 18.5.5. Application
- 18.5.6. End-Users
- 18.5.7. Imaging Guidance
- 18.5.8. Distribution Channel
- 18.6. Nigeria Cryoablation Devices Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Product Type
- 18.6.3. Technology
- 18.6.4. Therapy Type
- 18.6.5. Application
- 18.6.6. End-Users
- 18.6.7. Imaging Guidance
- 18.6.8. Distribution Channel
- 18.7. Algeria Cryoablation Devices Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Product Type
- 18.7.3. Technology
- 18.7.4. Therapy Type
- 18.7.5. Application
- 18.7.6. End-Users
- 18.7.7. Imaging Guidance
- 18.7.8. Distribution Channel
- 18.8. Rest of Africa Cryoablation Devices Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Product Type
- 18.8.3. Technology
- 18.8.4. Therapy Type
- 18.8.5. Application
- 18.8.6. End-Users
- 18.8.7. Imaging Guidance
- 18.8.8. Distribution Channel
- 19. South America Cryoablation Devices Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. South America Cryoablation Devices Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Product Type
- 19.3.2. Technology
- 19.3.3. Therapy Type
- 19.3.4. Application
- 19.3.5. End-Users
- 19.3.6. Imaging Guidance
- 19.3.7. Distribution Channel
- 19.3.8. Country
- 19.3.8.1. Brazil
- 19.3.8.2. Argentina
- 19.3.8.3. Rest of South America
- 19.4. Brazil Cryoablation Devices Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Product Type
- 19.4.3. Technology
- 19.4.4. Therapy Type
- 19.4.5. Application
- 19.4.6. End-Users
- 19.4.7. Imaging Guidance
- 19.4.8. Distribution Channel
- 19.5. Argentina Cryoablation Devices Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Product Type
- 19.5.3. Technology
- 19.5.4. Therapy Type
- 19.5.5. Application
- 19.5.6. End-Users
- 19.5.7. Imaging Guidance
- 19.5.8. Distribution Channel
- 19.6. Rest of South America Cryoablation Devices Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Product Type
- 19.6.3. Technology
- 19.6.4. Therapy Type
- 19.6.5. Application
- 19.6.6. End-Users
- 19.6.7. Imaging Guidance
- 19.6.8. Distribution Channel
- 20. Key Players/ Company Profile
- 20.1. Adagio Medical
- 20.1.1. Company Details/ Overview
- 20.1.2. Company Financials
- 20.1.3. Key Customers and Competitors
- 20.1.4. Business/ Industry Portfolio
- 20.1.5. Product Portfolio/ Specification Details
- 20.1.6. Pricing Data
- 20.1.7. Strategic Overview
- 20.1.8. Recent Developments
- 20.2. Apyx Medical Corporation
- 20.3. AtriCure
- 20.4. Boston Scientific Corporation
- 20.5. Brymill Cryogenic Systems
- 20.6. Channel Medsystem
- 20.7. CONMED Corporation
- 20.8. Cortex Technology
- 20.9. CryoConcepts LP
- 20.10. Cryomedix
- 20.11. CryoPen
- 20.12. Ellman International
- 20.13. Erbe Elektromedizin GmbH
- 20.14. Galil Medical
- 20.15. IceCure Medical
- 20.16. Medtronic
- 20.17. Metrum Cryoflex
- 20.18. OraSure Technologies
- 20.19. Sanarus Technologies
- 20.20. Stryker Corporation
- 20.21. Vascular Technology
- 20.22. Other Key Players
- 20.1. Adagio Medical
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
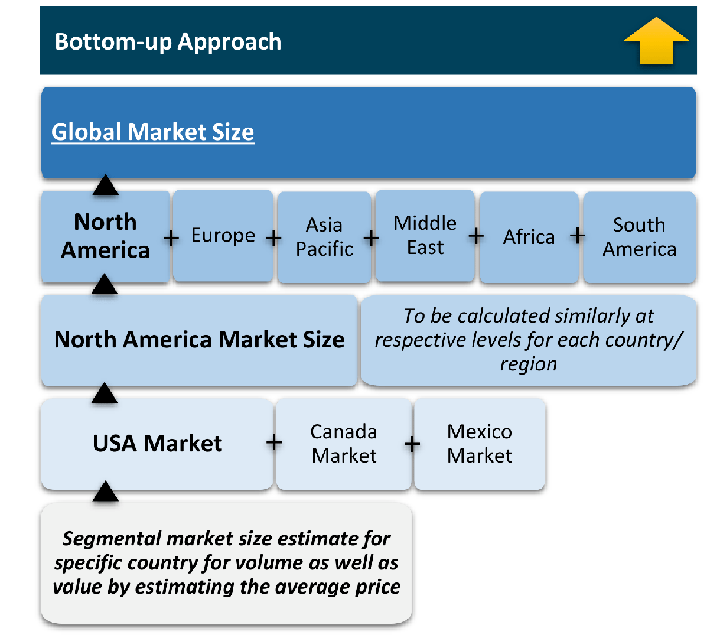
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation