Pharmaceutical Contract Manufacturing Market Size, Share & Trends Analysis Report by Service Type (API Manufacturing, Finished Dosage Form (FDF) Manufacturing, Packaging Services, Secondary Manufacturing, Clinical Trial Manufacturing, Commercial Manufacturing, Others), Drug Development Phase, Therapeutic Area, Drug Classification, Type of Contract, Packaging Type, End-users and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
Market Structure & Evolution |
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
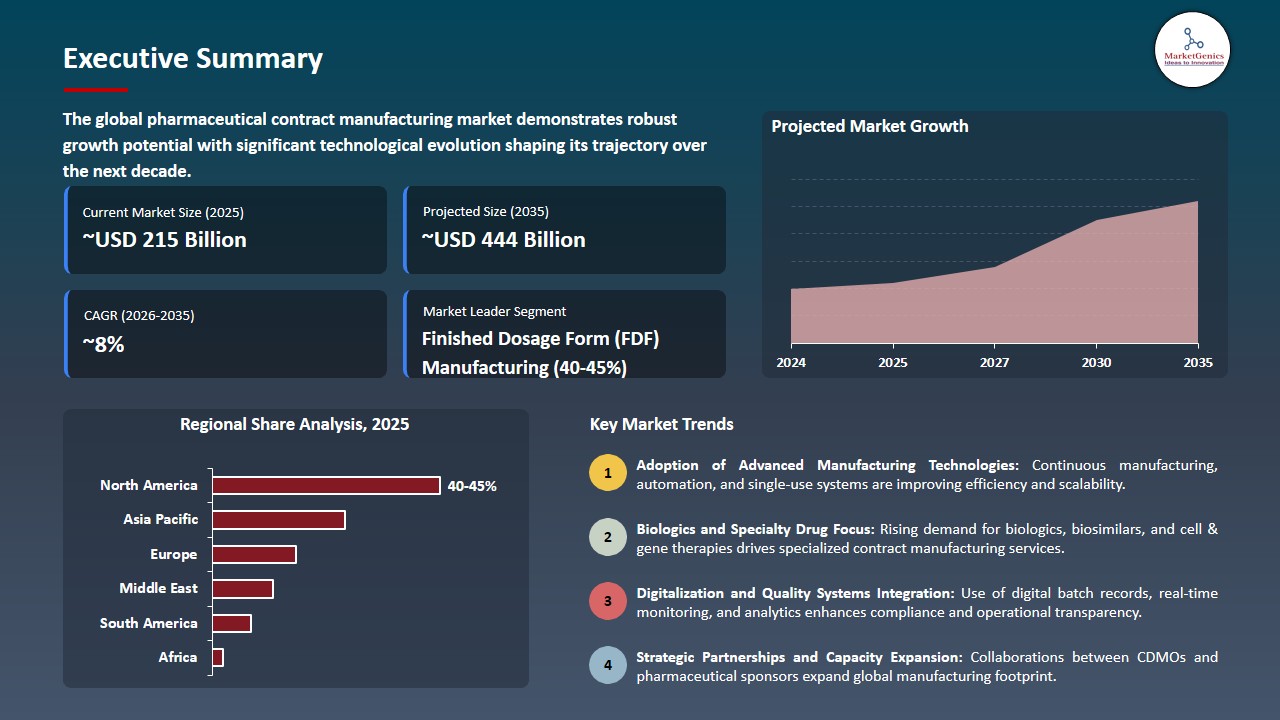
Pharmaceutical Contract Manufacturing Market Size, Share, and Growth
The global pharmaceutical contract manufacturing market is experiencing robust growth, with its estimated value of USD 215.3 billion in the year 2025 and USD 443.7 billion by 2035, registering a CAGR of 7.5% during the forecast period. The pharmaceutical contract manufacturing industry is experiencing exceptional growth on a global scale as pharmaceutical companies are increasingly outsourcing production.
Abzena and Argonaut Manufacturing Services have announced the formation of a new strategic partnership that will provide fully integrated manufacturing solutions for biopharmaceutical clients from early drug discovery through commercialized product. Officials from both companies indicated that the new partnership would facilitate a smooth transition from early drug discovery to commercial manufacturing.
Owing to increased complexity in drug development and companies are expanding their capabilities. For example, Samsung Biologics has expanded its capacity for large-scale production, while Lonza continues to invest heavily in biologics and advanced therapies due to high demand for production partners who provide reliable, compliant and scalable services.
The explosion in the market for biologic medicines, biosimilars, and specialty drugs has created a new demand for experienced contract manufacturers capable of meeting stringent regulatory and quality associated with all aspects of the production process. Regulatory agencies (such as the U.S. Food and Drug Administration and European Medicines Agency) have increased the level of regulatory oversight over contract manufacturers, resulting in increased partnerships between pharmaceutical companies and established contract manufacturers that provide validated facilities and global compliance expertise.
The combination of these factors, outsourcing, regulatory requirements, and need for complex therapeutic agents has led to significant growth of the overall market by allowing for rapid commercialization and enhanced supply chain resilience.
Contract manufacturers could add value across the pharmaceutical supply chain through provision of additional services to clients through adjacent services, such as active pharmaceutical ingredient manufacture, sterile fill/finish services, clinical trial material production, packaging and labelling solutions, and digital quality systems.

Pharmaceutical Contract Manufacturing Market Dynamics and Trends
Driver: Increasing Regulatory Stringency and Outsourcing Imperatives Driving Pharmaceutical Contract Manufacturing Adoption
-
The pharmaceutical industry's contracted manufacturers are growing rapidly as regulatory agencies around the world, including the U.S. Food and Drug Administration and The European Medicines Agency, are continually tightening quality, traceability and validation standards, encouraging drug developers find compliant contract development/manufacturing companies over creating their own in-house capability.
- A significant increase in the development of biologics, biosimilars and high potency drugs has increased the need for specialized contract manufacturers that have advanced containment measures, aseptic processing facilities and validated large scale manufacturing capabilities, and has consequently accelerated the trend towards outsourcing of both innovator and generic pharmaceuticals.
- The ongoing need to shorten the time required to take products to business and therefore reduce capital expenditure has resulted in a growing number of long-term manufacturing agreements/exclusivity between firms that develop both small molecular and biologic pipelines. All these factors are likely to boost the growth of the pharmaceutical contract manufacturing market.
Restraint: Capacity Constraints, Cost Pressures, and Technology Integration Challenges
-
The increased demand for biologics and complex therapies can now only be met if there is sufficient manufacturing capacity available, causing longer lead times and greater costs associated with contracts in relation to the target count of drugs being produced. For example, many collaborative biopharmaceutical organizations have recently cited extended lead times associated with production of biologics due to the inability of major contract development/ manufacturing organizations to produce large volumes of biologics.
- This pointed out that the industry continues to encounter significant technical bottlenecks in producing high volumes of biologics. Integrating contract manufacturers' systems/processes specific to the sponsor will require technological transfers, validations and regulatory approvals before contract manufacturers will be able to manufacture the drug or therapy.
- Because of this additional integration effort and the need to meet all applicable compliance requirements, companies must expect higher overall costs for their projects and an increase in the amount of time needed to complete their projects. All these elements are expected to restrict the expansion of the pharmaceutical contract manufacturing market.
Opportunity: Growth in Emerging Markets and Specialized Manufacturing Services
-
Opportunities from the continuing growth of the global pharmaceutical industry, combined with increasing capacity of Asian and Latin American contract manufacturers provide for the strengthening of regional manufacturers who want to serve customers worldwide while still complying with applicable international regulations.
- An example of how the production linked incentives program supports contract manufacturer investment in India is evident by the capacity expansion that has occurred at both active pharmaceutical ingredient and formulation manufacturers as a result of capacity growth and renewed investments in India".
- With increased demand for sterile fill-finish products, high-potency active pharmaceutical ingredients, and clinical trial supplies, all of which contract manufacturers can provide, these companies will be able to advance in the value chain due to support from governments promoting domestic pharmaceutical manufacturing and export competitiveness. All these elements are expected to create more opportunities for future in the pharmaceutical contract manufacturing market.
Key Trend: Adoption of Advanced Manufacturing Technologies and Digital Quality Systems
-
There is a growing use of continuous production, automated systems and digital quality management systems to improve efficiency and consistency and to comply with regulations in the manufacturing sector.
- For example, increasing use of manufacturing execution systems (MES) and electronic or digital batch records at top-tier contract manufacturers such as Thermo Fisher Scientific and Lonza, to provide for real-time monitoring of quality and for use in regulatory inspections. Data analytics, monitoring of events in near real-time, and elimination of paper-based forms for batch records are increasing the capacity for transparency and audit readiness within contract manufacturing businesses.
- The continuing trend of implementing technology-enabled, flexible manufacturing methods will change the landscape for pharmaceutical contract manufacturing on a global level. All these elements are expected to influence significant trends in the pharmaceutical contract manufacturing market.
Pharmaceutical Contract Manufacturing Market Analysis and Segmental Data
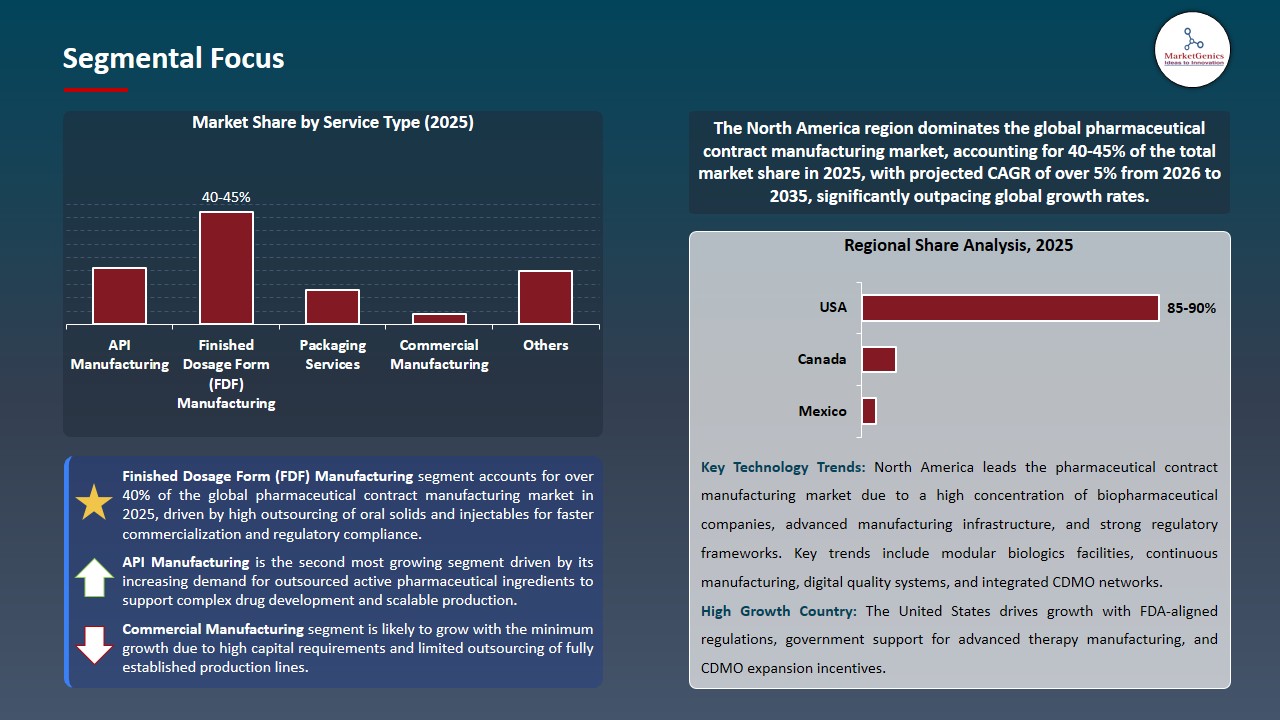
Finis Finished Dosage Form (FDF) Manufacturing Dominates the Global Pharmaceutical Contract Manufacturing Market
-
Manufacturing of finished pharmaceuticals is the major segment of contract manufacturing in the worldwide pharmaceuticals industry. This is due to highly complicated drug delivery systems, strict regulatory procedures required to get a final product approved and the need for scalable manufacturing systems that are ready for the market.
- Pharmaceutical companies are using contract manufacturers that have expertise in developed solid oral dosages, modified release formulations, sterile injectables and combination products that need an expert level of formulation and validated filling and finishing processes. Further, Serialization, labelling, and packaging compliance is increasing, thereby enhancing the demand for finished dosage products from contract manufacturers.
- For instance, Thermo Fisher Scientific recently adding to its sterile fill-finish manufacturing capacity in Europe and North America as part of its commercialization strategy for injectables, which emphasized the importance of having finished dosage capabilities in the contract manufacturing arena. Thus, the growth of the post-combustion is expected to continue in pharmaceutical contract manufacturing market globally.
North America Dominates the Pharmaceutical Contract Manufacturing Market amid Strong Regulatory Infrastructure and Biopharmaceutical Innovation
-
The North American pharmaceutical contract manufacturing market is characterized by a well-developed regulatory environment, high-quality manufacturing capabilities, and a large number of biopharmaceutical companies that invest in research & development. The early introduction of these new therapies in North America has contributed to the growth, particularly for specialized contract manufacturers who can provide compliant contract manufacturing for biologics, cell and gene therapies.
- North America's strong patent system, skilled labor, and proximity to large pharmaceutical companies further contribute to the attractiveness of outsourcing to contract development and manufacturing organizations within the region. An example of this continued investment in the North American contract manufacturing ecosystem is Novo Holdings recently acquiring Catalent for the purpose of enhancing the biological and advanced therapy contract manufacturing capacity in the US.
- Continued investment in research and development and positive funding environments provide additional impetus for the quicker progress of new products through clinical and commercial manufacturing development and production. Collectively, North America's leading position is expected to continue in the global pharmaceutical contract manufacturing market.
Pharmaceutical Contract Manufacturing Market Ecosystem
There is a moderately consolidated pharmaceutical contract manufacturing market, with Tier 1 companies like Thermo Fisher Scientific, Lonza and Samsung Biologics having a large share of the large biologicals and sterile manufacture business and Tier 2 and Tier 3 regional contract development and manufacturing organizations being a smaller segment of the market with niche and flexible, competitively priced services reflecting moderate fragmentation.
Key nodes of the pharmaceutical value-chain include API manufacture and FDF manufacture. An example of this is the recent expansion by Thermo Fisher Scientific in 2024-2025 of its U.S. sterile fill finish manufacturing capacity to enhance integration of downstream manufacture.

Recent Development and Strategic Overview:
-
In August 2025, Fujifilm Diosynth Biotechnologies announced plans to grow their large-scale production facilities for biologics in both the US and Europe, thereby enhancing end-to-end manufacturing services for monoclonal antibodies and recombinant proteins. Because, pharmaceutical companies are now able to benefit from integrated process development, commercial manufacture and regulatory backing all available through one global network, providing improved supply reliability and reduced time to market.
- In September 2024, WuXi Biologics took their global contract manufacturing platform to another level by adding modular single use bioreactor facilities across both Europe and Asia that support flexible scalable production of both biologics and biosimilars. This model helps accelerate technology transfers, lower capital risk for developers and enhance responsiveness to changes in clinical/commercial demand.
Report Scope
|
Attribute |
Detail |
|
Market Size in 2025 |
USD 215.3 Bn |
|
Market Forecast Value in 2035 |
USD 443.7 Bn |
|
Growth Rate (CAGR) |
7.5% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
USD Bn for Value |
|
Report Format |
Electronic (PDF) + Excel |
|
Regions and Countries Covered |
|||||
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Pharmaceutical Contract Manufacturing Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Pharmaceutical Contract Manufacturing Market, By Service Type |
|
|
Pharmaceutical Contract Manufacturing Market, By Drug Development Phase |
|
|
Pharmaceutical Contract Manufacturing Market, By Therapeutic Area |
|
|
Pharmaceutical Contract Manufacturing Market, By Drug Classification |
|
|
Pharmaceutical Contract Manufacturing Market, By Type of Contract |
|
|
Pharmaceutical Contract Manufacturing Market, By Packaging Type |
|
|
Pharmaceutical Contract Manufacturing Market, By End-users |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Pharmaceutical Contract Manufacturing Market Outlook
- 2.1.1. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Pharmaceutical Contract Manufacturing Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Industry Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.1. Global Healthcare & Pharmaceutical Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising outsourcing to reduce costs and accelerate drug commercialization.
- 4.1.1.2. Growing demand for biologics and complex formulations.
- 4.1.1.3. Capacity constraints in in-house manufacturing.
- 4.1.2. Restraints
- 4.1.2.1. Stringent regulatory and compliance requirements.
- 4.1.2.2. Quality and supply chain dependency risks.
- 4.1.2.3. High investment in advanced manufacturing technologies.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.5. Cost Structure Analysis
- 4.6. Porter’s Five Forces Analysis
- 4.7. PESTEL Analysis
- 4.8. Global Pharmaceutical Contract Manufacturing Market Demand
- 4.8.1. Historical Market Size – Value (US$ Bn), 2020-2024
- 4.8.2. Current and Future Market Size – Value (US$ Bn), 2026–2035
- 4.8.2.1. Y-o-Y Growth Trends
- 4.8.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Pharmaceutical Contract Manufacturing Market Analysis, by Service Type
- 6.1. Key Segment Analysis
- 6.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Service Type, 2021-2035
- 6.2.1. API Manufacturing
- 6.2.2. Finished Dosage Form (FDF) Manufacturing
- 6.2.3. Packaging Services
- 6.2.4. Secondary Manufacturing
- 6.2.5. Clinical Trial Manufacturing
- 6.2.6. Commercial Manufacturing
- 6.2.7. Others
- 7. Global Pharmaceutical Contract Manufacturing Market Analysis, by Drug Development Phase
- 7.1. Key Segment Analysis
- 7.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Drug Development Phase, 2021-2035
- 7.2.1. Preclinical Phase
- 7.2.2. Phase I Clinical Trials
- 7.2.3. Phase II Clinical Trials
- 7.2.4. Phase III Clinical Trials
- 7.2.5. Phase IV (Post-Marketing)
- 7.2.6. Commercial Production
- 8. Global Pharmaceutical Contract Manufacturing Market Analysis, by Therapeutic Area
- 8.1. Key Segment Analysis
- 8.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Therapeutic Area, 2021-2035
- 8.2.1. Oncology
- 8.2.2. Cardiovascular
- 8.2.3. Central Nervous System (CNS)
- 8.2.4. Diabetes & Metabolic Disorders
- 8.2.5. Respiratory Diseases
- 8.2.6. Infectious Diseases
- 8.2.7. Immunology & Inflammation
- 8.2.8. Gastrointestinal Disorders
- 8.2.9. Dermatology
- 8.2.10. Rare Diseases/Orphan Drugs
- 9. Global Pharmaceutical Contract Manufacturing Market Analysis, by Drug Classification
- 9.1. Key Segment Analysis
- 9.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Drug Classification, 2021-2035
- 9.2.1. Prescription Drugs
- 9.2.2. Over-the-Counter (OTC) Drugs
- 9.2.3. Generic Drugs
- 9.2.4. Branded Drugs
- 9.2.5. Specialty Drugs
- 9.2.6. Biosimilars
- 10. Global Pharmaceutical Contract Manufacturing Market Analysis, by Type of Contract
- 10.1. Key Segment Analysis
- 10.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Type of Contract, 2021-2035
- 10.2.1. Full-Service Contracts
- 10.2.2. Partial Service Contracts
- 10.2.3. Technology Transfer
- 10.2.4. Process Development
- 10.2.5. Manufacturing Only
- 11. Global Pharmaceutical Contract Manufacturing Market Analysis and Forecasts, by Packaging Type
- 11.1. Key Findings
- 11.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Packaging Type, 2021-2035
- 11.2.1. Primary Packaging
- 11.2.1.1. Blister Packs
- 11.2.1.2. Bottles
- 11.2.1.3. Vials & Ampoules
- 11.2.1.4. Sachets & Pouches
- 11.2.1.5. Others
- 11.2.2. Secondary Packaging
- 11.2.2.1. Cartons
- 11.2.2.2. Labels
- 11.2.2.3. Patient Information Leaflets
- 11.2.2.4. Others
- 11.2.3. Serialization & Track-and-Trace
- 11.2.1. Primary Packaging
- 12. Global Pharmaceutical Contract Manufacturing Market Analysis and Forecasts, by End-users
- 12.1. Key Findings
- 12.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by End-users, 2021-2035
- 12.2.1. Pharmaceutical Companies
- 12.2.2. Biotechnology Companies
- 12.2.3. Generic Drug Manufacturers
- 12.2.4. Specialty Pharmaceutical Companies
- 12.2.5. Virtual Pharma/Biotech Companies
- 12.2.6. Academic & Research Institutions
- 12.2.7. Contract Research Organizations (CROs)
- 12.2.8. Veterinary Pharmaceutical Companies
- 12.2.9. Others
- 13. Global Pharmaceutical Contract Manufacturing Market Analysis and Forecasts, by Region
- 13.1. Key Findings
- 13.2. Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 13.2.1. North America
- 13.2.2. Europe
- 13.2.3. Asia Pacific
- 13.2.4. Middle East
- 13.2.5. Africa
- 13.2.6. South America
- 14. North America Pharmaceutical Contract Manufacturing Market Analysis
- 14.1. Key Segment Analysis
- 14.2. Regional Snapshot
- 14.3. North America Pharmaceutical Contract Manufacturing Market Size Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 14.3.1. Service Type
- 14.3.2. Drug Development Phase
- 14.3.3. Therapeutic Area
- 14.3.4. Drug Classification
- 14.3.5. Type of Contract
- 14.3.6. Packaging Type
- 14.3.7. End-users
- 14.3.8. Country
- 14.3.8.1. USA
- 14.3.8.2. Canada
- 14.3.8.3. Mexico
- 14.4. USA Pharmaceutical Contract Manufacturing Market
- 14.4.1. Country Segmental Analysis
- 14.4.2. Service Type
- 14.4.3. Drug Development Phase
- 14.4.4. Therapeutic Area
- 14.4.5. Drug Classification
- 14.4.6. Type of Contract
- 14.4.7. Packaging Type
- 14.4.8. End-users
- 14.5. Canada Pharmaceutical Contract Manufacturing Market
- 14.5.1. Country Segmental Analysis
- 14.5.2. Service Type
- 14.5.3. Drug Development Phase
- 14.5.4. Therapeutic Area
- 14.5.5. Drug Classification
- 14.5.6. Type of Contract
- 14.5.7. Packaging Type
- 14.5.8. End-users
- 14.6. Mexico Pharmaceutical Contract Manufacturing Market
- 14.6.1. Country Segmental Analysis
- 14.6.2. Service Type
- 14.6.3. Drug Development Phase
- 14.6.4. Therapeutic Area
- 14.6.5. Drug Classification
- 14.6.6. Type of Contract
- 14.6.7. Packaging Type
- 14.6.8. End-users
- 15. Europe Pharmaceutical Contract Manufacturing Market Analysis
- 15.1. Key Segment Analysis
- 15.2. Regional Snapshot
- 15.3. Europe Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 15.3.1. Service Type
- 15.3.2. Drug Development Phase
- 15.3.3. Therapeutic Area
- 15.3.4. Drug Classification
- 15.3.5. Type of Contract
- 15.3.6. Packaging Type
- 15.3.7. End-users
- 15.3.8. Country
- 15.3.8.1. Germany
- 15.3.8.2. United Kingdom
- 15.3.8.3. France
- 15.3.8.4. Italy
- 15.3.8.5. Spain
- 15.3.8.6. Netherlands
- 15.3.8.7. Nordic Countries
- 15.3.8.8. Poland
- 15.3.8.9. Russia & CIS
- 15.3.8.10. Rest of Europe
- 15.4. Germany Pharmaceutical Contract Manufacturing Market
- 15.4.1. Country Segmental Analysis
- 15.4.2. Service Type
- 15.4.3. Drug Development Phase
- 15.4.4. Therapeutic Area
- 15.4.5. Drug Classification
- 15.4.6. Type of Contract
- 15.4.7. Packaging Type
- 15.4.8. End-users
- 15.5. United Kingdom Pharmaceutical Contract Manufacturing Market
- 15.5.1. Country Segmental Analysis
- 15.5.2. Service Type
- 15.5.3. Drug Development Phase
- 15.5.4. Therapeutic Area
- 15.5.5. Drug Classification
- 15.5.6. Type of Contract
- 15.5.7. Packaging Type
- 15.5.8. End-users
- 15.6. France Pharmaceutical Contract Manufacturing Market
- 15.6.1. Country Segmental Analysis
- 15.6.2. Service Type
- 15.6.3. Drug Development Phase
- 15.6.4. Therapeutic Area
- 15.6.5. Drug Classification
- 15.6.6. Type of Contract
- 15.6.7. Packaging Type
- 15.6.8. End-users
- 15.7. Italy Pharmaceutical Contract Manufacturing Market
- 15.7.1. Country Segmental Analysis
- 15.7.2. Service Type
- 15.7.3. Drug Development Phase
- 15.7.4. Therapeutic Area
- 15.7.5. Drug Classification
- 15.7.6. Type of Contract
- 15.7.7. Packaging Type
- 15.7.8. End-users
- 15.8. Spain Pharmaceutical Contract Manufacturing Market
- 15.8.1. Country Segmental Analysis
- 15.8.2. Service Type
- 15.8.3. Drug Development Phase
- 15.8.4. Therapeutic Area
- 15.8.5. Drug Classification
- 15.8.6. Type of Contract
- 15.8.7. Packaging Type
- 15.8.8. End-users
- 15.9. Netherlands Pharmaceutical Contract Manufacturing Market
- 15.9.1. Country Segmental Analysis
- 15.9.2. Service Type
- 15.9.3. Drug Development Phase
- 15.9.4. Therapeutic Area
- 15.9.5. Drug Classification
- 15.9.6. Type of Contract
- 15.9.7. Packaging Type
- 15.9.8. End-users
- 15.10. Nordic Countries Pharmaceutical Contract Manufacturing Market
- 15.10.1. Country Segmental Analysis
- 15.10.2. Service Type
- 15.10.3. Drug Development Phase
- 15.10.4. Therapeutic Area
- 15.10.5. Drug Classification
- 15.10.6. Type of Contract
- 15.10.7. Packaging Type
- 15.10.8. End-users
- 15.11. Poland Pharmaceutical Contract Manufacturing Market
- 15.11.1. Country Segmental Analysis
- 15.11.2. Service Type
- 15.11.3. Drug Development Phase
- 15.11.4. Therapeutic Area
- 15.11.5. Drug Classification
- 15.11.6. Type of Contract
- 15.11.7. Packaging Type
- 15.11.8. End-users
- 15.12. Russia & CIS Pharmaceutical Contract Manufacturing Market
- 15.12.1. Country Segmental Analysis
- 15.12.2. Service Type
- 15.12.3. Drug Development Phase
- 15.12.4. Therapeutic Area
- 15.12.5. Drug Classification
- 15.12.6. Type of Contract
- 15.12.7. Packaging Type
- 15.12.8. End-users
- 15.13. Rest of Europe Pharmaceutical Contract Manufacturing Market
- 15.13.1. Country Segmental Analysis
- 15.13.2. Service Type
- 15.13.3. Drug Development Phase
- 15.13.4. Therapeutic Area
- 15.13.5. Drug Classification
- 15.13.6. Type of Contract
- 15.13.7. Packaging Type
- 15.13.8. End-users
- 16. Asia Pacific Pharmaceutical Contract Manufacturing Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. Asia Pacific Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Service Type
- 16.3.2. Drug Development Phase
- 16.3.3. Therapeutic Area
- 16.3.4. Drug Classification
- 16.3.5. Type of Contract
- 16.3.6. Packaging Type
- 16.3.7. End-users
- 16.3.8. Country
- 16.3.8.1. China
- 16.3.8.2. India
- 16.3.8.3. Japan
- 16.3.8.4. South Korea
- 16.3.8.5. Australia and New Zealand
- 16.3.8.6. Indonesia
- 16.3.8.7. Malaysia
- 16.3.8.8. Thailand
- 16.3.8.9. Vietnam
- 16.3.8.10. Rest of Asia Pacific
- 16.4. China Pharmaceutical Contract Manufacturing Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Service Type
- 16.4.3. Drug Development Phase
- 16.4.4. Therapeutic Area
- 16.4.5. Drug Classification
- 16.4.6. Type of Contract
- 16.4.7. Packaging Type
- 16.4.8. End-users
- 16.5. India Pharmaceutical Contract Manufacturing Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Service Type
- 16.5.3. Drug Development Phase
- 16.5.4. Therapeutic Area
- 16.5.5. Drug Classification
- 16.5.6. Type of Contract
- 16.5.7. Packaging Type
- 16.5.8. End-users
- 16.6. Japan Pharmaceutical Contract Manufacturing Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Service Type
- 16.6.3. Drug Development Phase
- 16.6.4. Therapeutic Area
- 16.6.5. Drug Classification
- 16.6.6. Type of Contract
- 16.6.7. Packaging Type
- 16.6.8. End-users
- 16.7. South Korea Pharmaceutical Contract Manufacturing Market
- 16.7.1. Country Segmental Analysis
- 16.7.2. Service Type
- 16.7.3. Drug Development Phase
- 16.7.4. Therapeutic Area
- 16.7.5. Drug Classification
- 16.7.6. Type of Contract
- 16.7.7. Packaging Type
- 16.7.8. End-users
- 16.8. Australia and New Zealand Pharmaceutical Contract Manufacturing Market
- 16.8.1. Country Segmental Analysis
- 16.8.2. Service Type
- 16.8.3. Drug Development Phase
- 16.8.4. Therapeutic Area
- 16.8.5. Drug Classification
- 16.8.6. Type of Contract
- 16.8.7. Packaging Type
- 16.8.8. End-users
- 16.9. Indonesia Pharmaceutical Contract Manufacturing Market
- 16.9.1. Country Segmental Analysis
- 16.9.2. Service Type
- 16.9.3. Drug Development Phase
- 16.9.4. Therapeutic Area
- 16.9.5. Drug Classification
- 16.9.6. Type of Contract
- 16.9.7. Packaging Type
- 16.9.8. End-users
- 16.10. Malaysia Pharmaceutical Contract Manufacturing Market
- 16.10.1. Country Segmental Analysis
- 16.10.2. Service Type
- 16.10.3. Drug Development Phase
- 16.10.4. Therapeutic Area
- 16.10.5. Drug Classification
- 16.10.6. Type of Contract
- 16.10.7. Packaging Type
- 16.10.8. End-users
- 16.11. Thailand Pharmaceutical Contract Manufacturing Market
- 16.11.1. Country Segmental Analysis
- 16.11.2. Service Type
- 16.11.3. Drug Development Phase
- 16.11.4. Therapeutic Area
- 16.11.5. Drug Classification
- 16.11.6. Type of Contract
- 16.11.7. Packaging Type
- 16.11.8. End-users
- 16.12. Vietnam Pharmaceutical Contract Manufacturing Market
- 16.12.1. Country Segmental Analysis
- 16.12.2. Service Type
- 16.12.3. Drug Development Phase
- 16.12.4. Therapeutic Area
- 16.12.5. Drug Classification
- 16.12.6. Type of Contract
- 16.12.7. Packaging Type
- 16.12.8. End-users
- 16.13. Rest of Asia Pacific Pharmaceutical Contract Manufacturing Market
- 16.13.1. Country Segmental Analysis
- 16.13.2. Service Type
- 16.13.3. Drug Development Phase
- 16.13.4. Therapeutic Area
- 16.13.5. Drug Classification
- 16.13.6. Type of Contract
- 16.13.7. Packaging Type
- 16.13.8. End-users
- 17. Middle East Pharmaceutical Contract Manufacturing Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Middle East Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Service Type
- 17.3.2. Drug Development Phase
- 17.3.3. Therapeutic Area
- 17.3.4. Drug Classification
- 17.3.5. Type of Contract
- 17.3.6. Packaging Type
- 17.3.7. End-users
- 17.3.8. Country
- 17.3.8.1. Turkey
- 17.3.8.2. UAE
- 17.3.8.3. Saudi Arabia
- 17.3.8.4. Israel
- 17.3.8.5. Rest of Middle East
- 17.4. Turkey Pharmaceutical Contract Manufacturing Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Service Type
- 17.4.3. Drug Development Phase
- 17.4.4. Therapeutic Area
- 17.4.5. Drug Classification
- 17.4.6. Type of Contract
- 17.4.7. Packaging Type
- 17.4.8. End-users
- 17.5. UAE Pharmaceutical Contract Manufacturing Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Service Type
- 17.5.3. Drug Development Phase
- 17.5.4. Therapeutic Area
- 17.5.5. Drug Classification
- 17.5.6. Type of Contract
- 17.5.7. Packaging Type
- 17.5.8. End-users
- 17.6. Saudi Arabia Pharmaceutical Contract Manufacturing Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Service Type
- 17.6.3. Drug Development Phase
- 17.6.4. Therapeutic Area
- 17.6.5. Drug Classification
- 17.6.6. Type of Contract
- 17.6.7. Packaging Type
- 17.6.8. End-users
- 17.7. Israel Pharmaceutical Contract Manufacturing Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Service Type
- 17.7.3. Drug Development Phase
- 17.7.4. Therapeutic Area
- 17.7.5. Drug Classification
- 17.7.6. Type of Contract
- 17.7.7. Packaging Type
- 17.7.8. End-users
- 17.8. Rest of Middle East Pharmaceutical Contract Manufacturing Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Service Type
- 17.8.3. Drug Development Phase
- 17.8.4. Therapeutic Area
- 17.8.5. Drug Classification
- 17.8.6. Type of Contract
- 17.8.7. Packaging Type
- 17.8.8. End-users
- 18. Africa Pharmaceutical Contract Manufacturing Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Africa Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Service Type
- 18.3.2. Drug Development Phase
- 18.3.3. Therapeutic Area
- 18.3.4. Drug Classification
- 18.3.5. Type of Contract
- 18.3.6. Packaging Type
- 18.3.7. End-users
- 18.3.8. Country
- 18.3.8.1. South Africa
- 18.3.8.2. Egypt
- 18.3.8.3. Nigeria
- 18.3.8.4. Algeria
- 18.3.8.5. Rest of Africa
- 18.4. South Africa Pharmaceutical Contract Manufacturing Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Service Type
- 18.4.3. Drug Development Phase
- 18.4.4. Therapeutic Area
- 18.4.5. Drug Classification
- 18.4.6. Type of Contract
- 18.4.7. Packaging Type
- 18.4.8. End-users
- 18.5. Egypt Pharmaceutical Contract Manufacturing Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Service Type
- 18.5.3. Drug Development Phase
- 18.5.4. Therapeutic Area
- 18.5.5. Drug Classification
- 18.5.6. Type of Contract
- 18.5.7. Packaging Type
- 18.5.8. End-users
- 18.6. Nigeria Pharmaceutical Contract Manufacturing Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Service Type
- 18.6.3. Drug Development Phase
- 18.6.4. Therapeutic Area
- 18.6.5. Drug Classification
- 18.6.6. Type of Contract
- 18.6.7. Packaging Type
- 18.6.8. End-users
- 18.7. Algeria Pharmaceutical Contract Manufacturing Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Service Type
- 18.7.3. Drug Development Phase
- 18.7.4. Therapeutic Area
- 18.7.5. Drug Classification
- 18.7.6. Type of Contract
- 18.7.7. Packaging Type
- 18.7.8. End-users
- 18.8. Rest of Africa Pharmaceutical Contract Manufacturing Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Service Type
- 18.8.3. Drug Development Phase
- 18.8.4. Therapeutic Area
- 18.8.5. Drug Classification
- 18.8.6. Type of Contract
- 18.8.7. Packaging Type
- 18.8.8. End-users
- 19. South America Pharmaceutical Contract Manufacturing Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. South America Pharmaceutical Contract Manufacturing Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Service Type
- 19.3.2. Drug Development Phase
- 19.3.3. Therapeutic Area
- 19.3.4. Drug Classification
- 19.3.5. Type of Contract
- 19.3.6. Packaging Type
- 19.3.7. End-users
- 19.3.8. Country
- 19.3.8.1. Brazil
- 19.3.8.2. Argentina
- 19.3.8.3. Rest of South America
- 19.4. Brazil Pharmaceutical Contract Manufacturing Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Service Type
- 19.4.3. Drug Development Phase
- 19.4.4. Therapeutic Area
- 19.4.5. Drug Classification
- 19.4.6. Type of Contract
- 19.4.7. Packaging Type
- 19.4.8. End-users
- 19.5. Argentina Pharmaceutical Contract Manufacturing Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Service Type
- 19.5.3. Drug Development Phase
- 19.5.4. Therapeutic Area
- 19.5.5. Drug Classification
- 19.5.6. Type of Contract
- 19.5.7. Packaging Type
- 19.5.8. End-users
- 19.6. Rest of South America Pharmaceutical Contract Manufacturing Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Service Type
- 19.6.3. Drug Development Phase
- 19.6.4. Therapeutic Area
- 19.6.5. Drug Classification
- 19.6.6. Type of Contract
- 19.6.7. Packaging Type
- 19.6.8. End-users
- 20. Key Players/ Company Profile
- 20.1. AbbVie Contract Manufacturing
- 20.1.1. Company Details/ Overview
- 20.1.2. Company Financials
- 20.1.3. Key Customers and Competitors
- 20.1.4. Business/ Industry Portfolio
- 20.1.5. Product Portfolio/ Specification Details
- 20.1.6. Pricing Data
- 20.1.7. Strategic Overview
- 20.1.8. Recent Developments
- 20.2. Adare Pharmaceuticals
- 20.3. Aenova Group
- 20.4. Alcami Corporation
- 20.5. Boehringer Ingelheim BioXcellence
- 20.6. Catalent Pharmaceuticals
- 20.7. Consort Medical (Recipharm)
- 20.8. Dishman Carbogen Amcis
- 20.9. Famar Health Care Services
- 20.10. Fareva
- 20.11. Lonza Group
- 20.12. Metrics Contract Services
- 20.13. Millmount Healthcare
- 20.14. Nephron Pharmaceuticals
- 20.15. Patheon (Part of Thermo Fisher)
- 20.16. Piramal Pharma Solutions
- 20.17. Recipharm
- 20.18. Samsung Biologics
- 20.19. Siegfried Holding
- 20.20. Thermo Fisher Scientific (Patheon)
- 20.21. Vetter Pharma International
- 20.22. Other Key Players
- 20.1. AbbVie Contract Manufacturing
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
Research Design
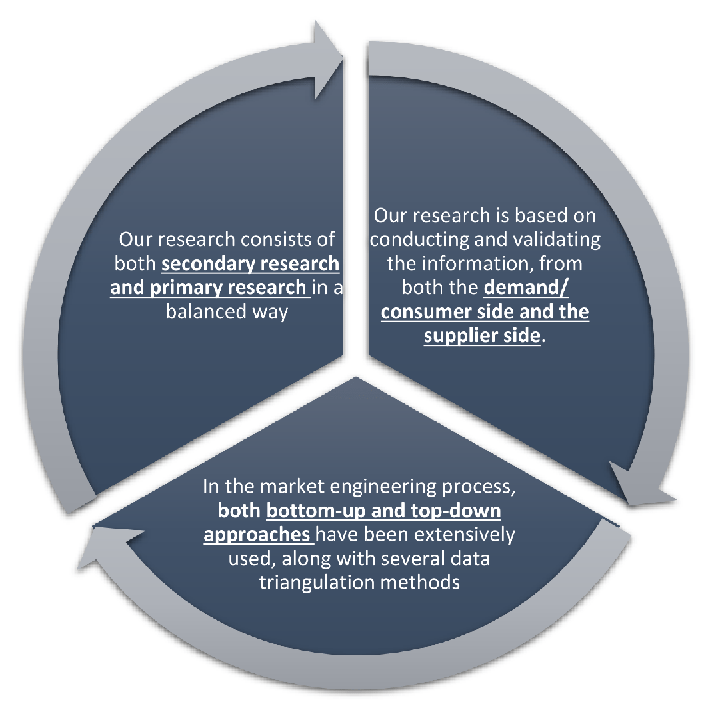
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
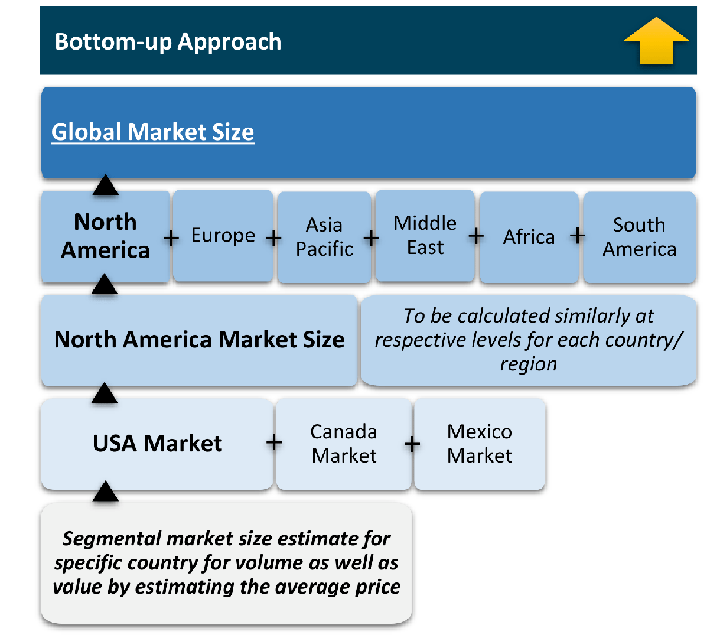
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation