Point of Care Testing Market Size, Share & Trends Analysis Report by Product Type (Glucose Testing Kits, Infectious Disease Testing Kits, Cardiac Marker Testing Kits, Pregnancy and Fertility Testing Kits, Blood Gas/Electrolyte Testing Kits, Cholesterol Testing Kits, Others), Technology Platform, Sample Type, Mode of Purchase, Testing Complexity, Test Result Time, Number of Parameters Tested, Connectivity, End-use and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Point of Care Testing Market Size, Share, and Growth
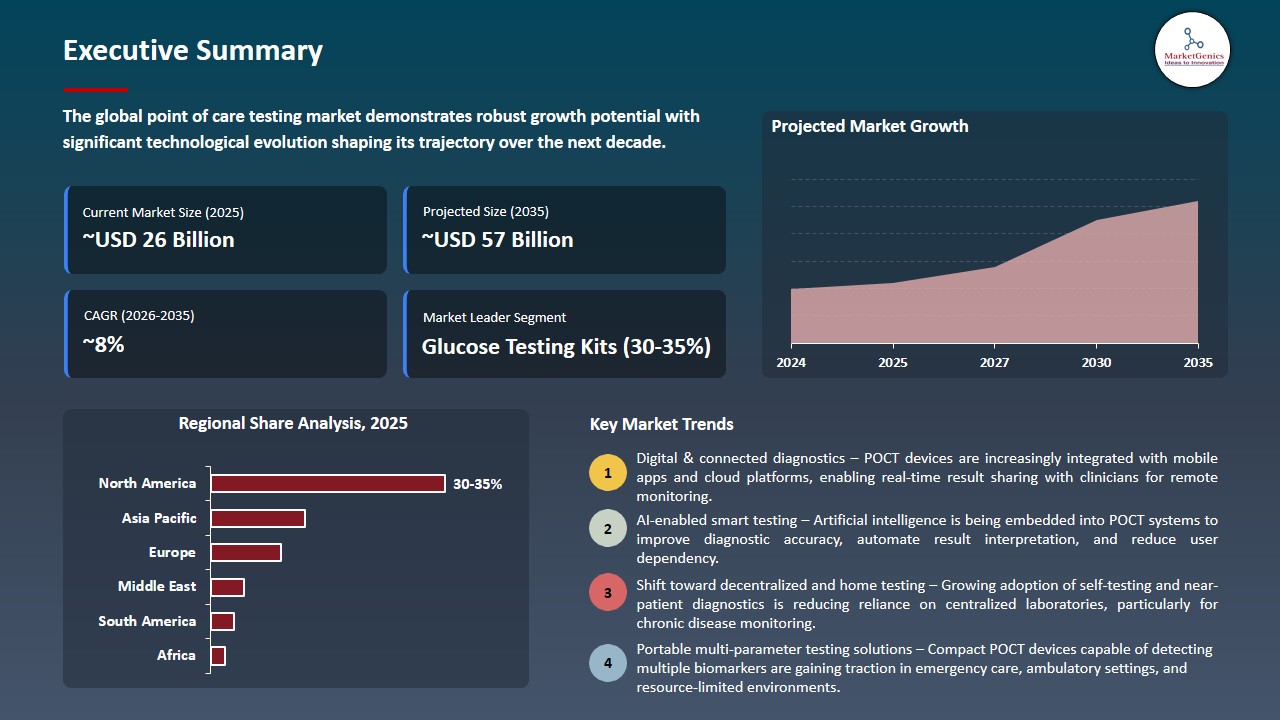
The global point of care testing market is witnessing strong growth, valued at USD 26.4 billion in 2025 and projected to reach USD 56.5 billion by 2035, expanding at a CAGR of 7.9% during the forecast period. Asia Pacific is the fastest-growing region in the point of care testing market due to rising chronic disease prevalence, expanding healthcare access, increasing adoption of rapid diagnostics, and supportive government initiatives to strengthen decentralized healthcare delivery.
Dwight Egan, CEO of Co-Diagnostics, said, “We are pleased with the diligent work of our development team in preparing this test to begin this clinical performance testing, Regulatory clearance and commercialization of the Co-Dx PCR platform remain the Company's primary objectives, and our research suggests that the market is primed for decentralized, gold-standard PCR testing, especially for respiratory infections. Our platform is designed to meet this growing demand by bringing accurate, reliable testing options to near-patient environments”.
The increased incidence of chronic and infectious diseases forms a major force behind the point of care testing market because diseases like diabetes, cardiovascular diseases and respiratory diseases are subject to regular testing and frequent monitoring. POCT allows real-time and fast diagnostic outcomes at and or close to the location of the patient, assists in making timely clinical decisions, enhancing disease management, and decreasing the reliance on centralized labs in a variety of healthcare settings.
The point of care testing market has a great growth potential with the integration of digital and telehealth synergy. The implementation of interconnected POCT devices combined with mobile applications and cloud platform facilitates a free flow of data, remote follow-up, and real-time clinical supervision. The integration also increases patient engagement, facilitates decentralized care models, and adds continuity of care, particularly in regard to chronic disease management and home-based testing settings.
Adjacent market opportunities in point of care testing are the growth of at-home testing kits, the growth of POCT in pharmacies and ambulatory care centers, the growing need to screen infectious diseases in the shortest possible time during outbreaks, increased use in aging populations and long-term care centers, and the advancement of multiplex and minimally invasive tests that enhance the efficiency, patient comfort, and clinical workflow across the decentralized care setting.

Point of Care Testing Market Dynamics and Trends
Driver: Need for rapid diagnostics and immediate results
-
The increasing demand of the fast diagnostics and immediate outcome is a significant impetus of the point of care testing market. POCT also has a great impact on the turnaround time, giving test results at or close to the patient site and the delay incurred in transporting the samples to the central laboratory party is eliminated.
- Quicker outcomes allow clinical decisions to be made in a timely manner, treatment to be initiated early, and better patient outcomes especially in emergency care, critical care, and handling infectious diseases.
- Co-Diagnostics, Co-Dx PCR Flu A/B, COVID-19, RSV Test Kit, on the Co-Dx PCR Pro platform, was initiated in clinical testing in November 2025, highlighting the increased need to decentralize rapid diagnostics. The point-of-care multiplex PCR test has been developed to provide patients with correct and near-patient results of respiratory infections, and make clinical decisions more rapidly.
- The speed also improves the efficiency of workflow by healthcare providers, reduces hospitalization, and facilitates efficient use of triage and disease monitoring in various healthcare environments.
Restraint: Quality, accuracy, and standardization concerns
-
Quality, accuracy, and standardization issues are also one of the significant limitations within the point of care testing market because the results of tests can be different when compared to centralized laboratory diagnostics. Variability may be due to the difference in the calibration of the device used, the quality of the reagent, the expertise of the operator and the environment in which the test is conducted that may influence the accuracy of analysis and clinical reliability of the test especially in the case of complex or critical diagnoses.
- Moreover, the absence of consistent standards across the point of care testing platforms presents the difficulties of consistency and comparability of results across different health care environments. Decentralized testing environments tend to be limited in terms of quality assurance controls, employee training and regulation.
- These elements may lead to higher risks of making diagnostic errors and low clinician confidence, which ultimately can slow the overall adoption of point of care testing in an environment where high precision and standardized performance are needed.
Opportunity: Broader POC test menu and multiplex testing
-
The growth of larger point-of-care test menus and multiplex testing is an important opportunity to the point of care testing market. The current trends of assay design and detection technology allow conducting multiplex tests, which can identify several pathogens or biomarkers in the same sample, enhancing the efficiency of diagnostics and the clinical process.
- The incorporation of high-sensitivity biomarkers can help identify the disease sooner and monitor more accurately the complicated diseases. In January 2025, Roche was given FDA 510(k) clearance, and CLIA waiver of cobas liat STI multiplex assays (CT/NG and CT/NG/MG panels) on Roche. These are exemplified by high sensitivity levels of PCR technology activated in these rapid molecular point of care tests to detect and distinguish several sexually transmitted infections with the use of one sample and give results within minutes.
- These innovations improve clinical value of point of care testing in the emergency care, infectious disease management, chronic disease monitoring, and decrease the time, cost and sample volume requirement in the decentralized care setting.
Key Trend: Digital & AI-enabled point of care systems
-
Digital and AI-enabled point-of-care systems are another important trend of the point of care testing market, which is based on the combination of smart devices with artificial intelligence (AI) and machine learning (ML) features. These sophisticated systems are capable of real time processing of complex data giving higher diagnostic accuracy, predictive information and clinical decision support.
- AI-based POCT devices are used to detect trends, identify abnormalities, and provide individualized advice, minimizing diagnostic inaccuracies and enhancing patient care. With digital connectivity, cloud-based applications, and AI analytics converging, the sharing of data becomes also seamless, the remote monitoring is also possible, and it is also possible to integrate with electronic health records, which facilitates efficient, decentralized care delivery.
- In 2025, the Mobilab, an Indian developer of point-of-care blood testing devices, backed by India’s Technology Development Board (TDB), is an IoT-enabled, AI/ML-powered system called the Mobilab. The portable analyzer which can test more than 25 parameters, kidney, liver, heart, vitamins, and cancer are some of them, incorporates AI algorithms to improve the accuracy and efficiency of the diagnosis.
- These developments highlight the importance of efficient AI-based, connected POCT systems in providing quicker, more precise, and available diagnostics in decentralized health care environments.
Point-of-Care-Testing-Market Analysis and Segmental Data
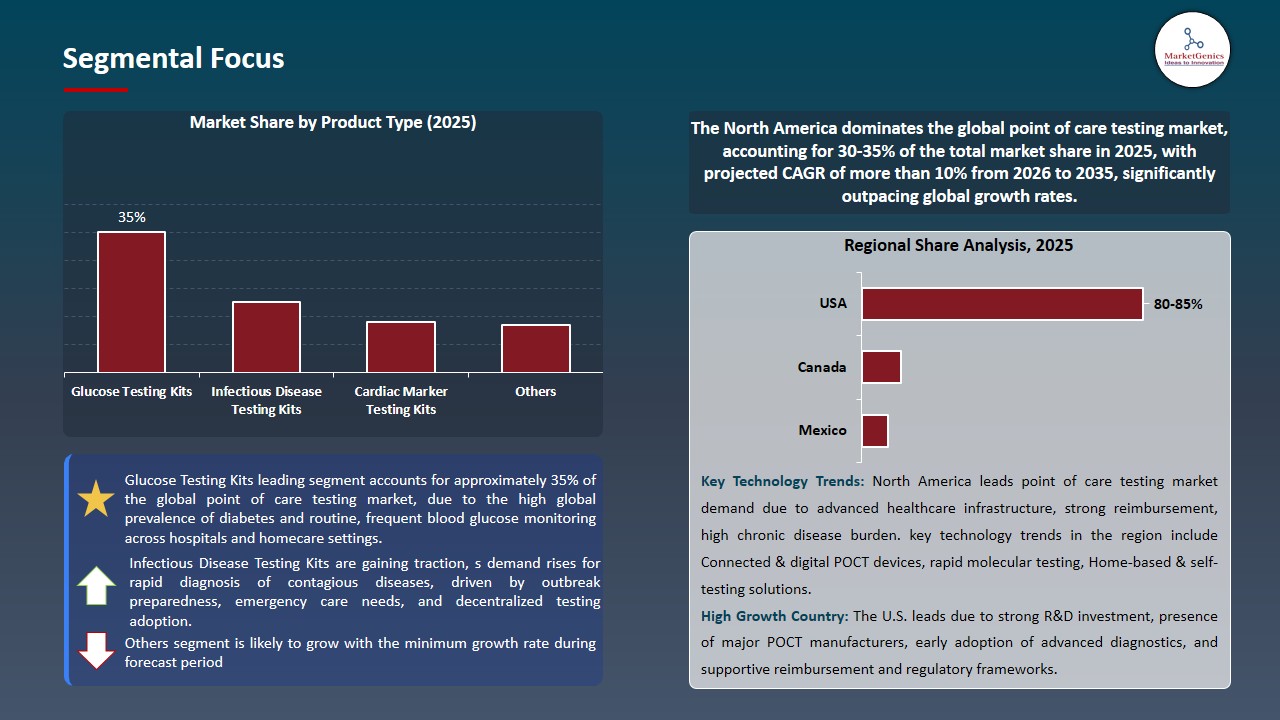
Glucose Testing Kits Dominate Global Point of Care Testing Market
-
Glucose testing kits remain the leading segment in the global point of care testing market, accounting for the largest share due to the rising prevalence of diabetes and increasing awareness of glycemic monitoring. These kits offer rapid, convenient, and reliable blood glucose measurement, enabling patients and healthcare providers to make timely treatment decisions.
- The ease of use, portability, and affordability of glucose meters have fueled their adoption across hospitals, clinics, and home-care settings. Additionally, advancements in continuous glucose monitoring systems and integration with mobile apps for real-time tracking further strengthen their market dominance, making glucose testing one of the most widely utilized and essential applications in the point of care testing landscape.
- In August 2025, Abbott launched the FreeStyle Libre 2 Plus in India, a continuous glucose monitoring (CGM) system that delivers automatic, real-time glucose readings to smartphones with optional high and low alerts. Designed for point-of-care use, the device enables people with diabetes and caregivers to manage the condition confidently without routine fingersticks, providing rapid, accurate, and actionable data.
- Glucose testing kits remain a cornerstone of the point of care testing market, driving widespread adoption and shaping the growth of decentralized diagnostics.
North America Leads Global Point of Care Testing Market Demand
-
North America is the leading region in the global point of care testing market, driven by a high prevalence of chronic and acute health conditions that require rapid diagnostics and continuous monitoring. Widespread awareness of diseases such as diabetes, cardiovascular disorders, and respiratory infections has increased demand for convenient, near-patient testing solutions, enabling timely clinical decision-making and improved patient outcomes.
- The region benefits from advanced healthcare infrastructure, well-established diagnostic laboratories, and a strong network of skilled healthcare professionals, which facilitate the adoption of innovative point of care testing technologies. Favorable reimbursement policies and insurance coverage further reduce financial barriers, encouraging hospitals, clinics, and home-care providers to integrate point-of-care solutions into routine practice.
- Additionally, North America is at the forefront of technological advancements in point of care testing, including multiplex assays, high-sensitivity biomarker detection, and AI-enabled diagnostic systems.
- These innovations, combined with a patient-centric focus on convenience and real-time monitoring, reinforce North America’s position as the leading regional market and a key driver of global point of care testing growth.
Point-of-Care-Testing-Market Ecosystem
The global point of care testing market is moderately fragmented, with key players including Abbott Laboratories, Roche Diagnostics, Danaher Corporation, Becton, Dickinson and Company (BD), and Siemens Healthineers. These companies maintain competitive strength through significant investments in research and development, innovative diagnostic technologies, robust product portfolios, and strong relationships with healthcare providers, clinics, and hospitals. Their market presence is further reinforced by physician training programs, regulatory expertise, and growing adoption of decentralized testing solutions across diverse clinical settings.
The point of care testing value chain encompasses raw material procurement and component manufacturing, device design and assembly under strict quality and safety standards, clinical validation and regulatory approvals, product branding and packaging, and distribution through hospitals, clinics, and home-care channels. Post-market activities include physician and patient education, remote monitoring, device maintenance, and therapy optimization.
Entry barriers in the point of care testing market remain high due to stringent regulatory requirements, the need for extensive clinical validation, technological complexity, and strong competition from established players. Continuous innovation in multiplex assays, AI-enabled diagnostics, miniaturized devices, and connected platforms drives differentiation and adoption across the global point of care testing landscape.

Recent Development and Strategic Overview:
-
In July 2025, Becton, Dickinson and Company (BD) received FDA 510(k) clearance for the BD Veritor System for SARS-CoV-2, a rapid, CLIA-waived point-of-care antigen test delivering digital results in about 15 minutes. The test enables timely COVID-19 detection in clinics, urgent care centers, and retail healthcare settings, supporting faster clinical decision-making.
- In June 2025, India’s ICMR-National Institute of Immunohaematology (NIIH) developed a simple point-of-care test for Haemophilia A and Von Willebrand Disease, enabling rapid, on-site detection of these bleeding disorders. The test provides a cost-effective, accessible diagnostic option for early disease identification and management, particularly in decentralized and resource-limited healthcare settings.
Report Scope
|
Detail |
|
|
Market Size in 2025 |
USD 26.4 Bn |
|
Market Forecast Value in 2035 |
USD 56.5 Bn |
|
Growth Rate (CAGR) |
7.9% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
US$ Billion for Value Thousand Units for Volume |
|
Report Format |
Electronic (PDF) + Excel |
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Point-of-Care-Testing-Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Point of Care Testing Market, By Product Type |
|
|
Point of Care Testing Market, By Technology Platform |
|
|
Point of Care Testing Market, By Sample Type |
|
|
Point of Care Testing Market, By Mode of Purchase |
|
|
Point of Care Testing Market, By Testing Complexity |
|
|
Point of Care Testing Market, By Test Result Time |
|
|
Point of Care Testing Market, By Number of Parameters Tested |
|
|
Point of Care Testing Market, By Connectivity |
|
|
Point of Care Testing Market, By End-use |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Point of Care Testing Market Outlook
- 2.1.1. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Point of Care Testing Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Industry Ecosystem Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.4.1. Import & Export Analysis, 2025
- 3.4.2. Top Importing Countries
- 3.4.3. Top Exporting Countries
- 3.5. Trump Tariff Impact Analysis
- 3.5.1. Manufacturer
- 3.5.1.1. Based on the component & Raw material
- 3.5.2. Supply Chain
- 3.5.3. End Consumer
- 3.5.1. Manufacturer
- 3.6. Raw Material Analysis
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising prevalence of chronic and infectious diseases increasing demand for rapid, near-patient diagnostics
- 4.1.1.2. Technological advancements enhancing accuracy, portability, and ease of use of point-of-care testing devices
- 4.1.1.3. Growing shift toward decentralized healthcare, home testing, and remote patient monitoring
- 4.1.2. Restraints
- 4.1.2.1. Stringent regulatory requirements and standardization challenges for product approvals
- 4.1.2.2. High costs of advanced point-of-care devices and limited reimbursement in some regions
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Ecosystem Analysis
- 4.5. Porter’s Five Forces Analysis
- 4.6. PESTEL Analysis
- 4.7. Global Point of Care Testing Market Demand
- 4.7.1. Historical Market Size – Volume (Thousand Units) and Value (US$ Bn), 2020-2024
- 4.7.2. Current and Future Market Size – Volume (Thousand Units) and Value (US$ Bn), 2026–2035
- 4.7.2.1. Y-o-Y Growth Trends
- 4.7.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Point of Care Testing Market Analysis, by Product Type
- 6.1. Key Segment Analysis
- 6.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Product Type, 2021-2035
- 6.2.1. Glucose Testing Kits
- 6.2.2. Infectious Disease Testing Kits
- 6.2.3. Cardiac Marker Testing Kits
- 6.2.4. Pregnancy and Fertility Testing Kits
- 6.2.5. Blood Gas/Electrolyte Testing Kits
- 6.2.6. Cholesterol Testing Kits
- 6.2.7. Others
- 7. Global Point of Care Testing Market Analysis, by Technology Platform
- 7.1. Key Segment Analysis
- 7.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Technology Platform, 2021-2035
- 7.2.1. Lateral Flow Assays
- 7.2.2. Immunoassays
- 7.2.3. Molecular Diagnostics
- 7.2.3.1. PCR-based
- 7.2.3.2. Isothermal Amplification
- 7.2.3.3. Microarray-based
- 7.2.3.4. Others
- 7.2.4. Clinical Chemistry
- 7.2.5. Hematology
- 7.2.6. Dipstick Technology
- 7.2.7. Microfluidics
- 7.2.8. Biosensors
- 7.2.8.1. Electrochemical Biosensors
- 7.2.8.2. Optical Biosensors
- 7.2.8.3. Piezoelectric Biosensors
- 7.2.8.4. Others
- 7.2.9. Other Technologies
- 8. Global Point of Care Testing Market Analysis, by Sample Type
- 8.1. Key Segment Analysis
- 8.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Sample Type, 2021-2035
- 8.2.1. Blood Samples
- 8.2.1.1. Whole Blood
- 8.2.1.2. Plasma
- 8.2.1.3. Serum
- 8.2.2. Urine Samples
- 8.2.3. Saliva Samples
- 8.2.4. Fecal Samples
- 8.2.5. Nasal/Throat Swabs
- 8.2.6. Other Body Fluids
- 8.2.1. Blood Samples
- 9. Global Point of Care Testing Market Analysis, by Mode of Purchase
- 9.1. Key Segment Analysis
- 9.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Implant Material, 2021-2035
- 9.2.1. Prescription-based Devices
- 9.2.2. Over-the-Counter (OTC) Devices
- 10. Global Point of Care Testing Market Analysis, by Testing Complexity
- 10.1. Key Segment Analysis
- 10.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Testing Complexity, 2021-2035
- 10.2.1. CLIA-Waived Tests
- 10.2.2. Moderate Complexity Tests
- 10.2.3. High Complexity Tests
- 11. Global Point of Care Testing Market Analysis, by Test Result Time
- 11.1. Key Segment Analysis
- 11.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Test Result Time, 2021-2035
- 11.2.1. Rapid Tests (<15 minutes)
- 11.2.2. Semi-Rapid Tests (15-30 minutes)
- 11.2.3. Standard Tests (>30 minutes)
- 12. Global Point of Care Testing Market Analysis and Forecasts, by Number of Parameters Tested
- 12.1. Key Findings
- 12.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Number of Parameters Tested, 2021-2035
- 12.2.1. Single-Parameter Tests
- 12.2.2. Multi-Parameter Tests
- 12.2.2.1. 2-5 Parameters
- 12.2.2.2. 6-10 Parameters
- 12.2.2.3. More than 10 Parameters
- 13. Global Point of Care Testing Market Analysis and Forecasts, by Connectivity
- 13.1. Key Findings
- 13.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Connectivity, 2021-2035
- 13.2.1. Standalone Devices
- 13.2.2. Connected Devices
- 13.2.2.1. Bluetooth-enabled
- 13.2.2.2. WiFi-enabled
- 13.2.2.3. Cloud-connected
- 13.2.3. Integrated Laboratory Systems
- 14. Global Point of Care Testing Market Analysis and Forecasts, by End-use
- 14.1. Key Findings
- 14.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by End-use, 2021-2035
- 14.2.1. Ambulatory Surgical Centers
- 14.2.2. Clinics
- 14.2.3. Diagnostic Laboratories
- 14.2.4. Home Care Settings
- 14.2.5. Hospitals
- 14.2.6. Military/Defense
- 14.2.7. Nursing Homes/Assisted Living
- 14.2.8. Pharmacies/Retail Clinics
- 14.2.9. Research Laboratories
- 14.2.10. Workplace/Occupational Health
- 14.2.11. Others
- 15. Global Point of Care Testing Market Analysis and Forecasts, by Region
- 15.1. Key Findings
- 15.2. Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 15.2.1. North America
- 15.2.2. Europe
- 15.2.3. Asia Pacific
- 15.2.4. Middle East
- 15.2.5. Africa
- 15.2.6. South America
- 16. North America Point of Care Testing Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. North America Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Product Type
- 16.3.2. Technology Platform
- 16.3.3. Sample Type
- 16.3.4. Mode of Purchase
- 16.3.5. Testing Complexity
- 16.3.6. Test Result Time
- 16.3.7. Connectivity
- 16.3.8. End-Use
- 16.3.9. Country
- 16.3.9.1. USA
- 16.3.9.2. Canada
- 16.3.9.3. Mexico
- 16.4. USA Point of Care Testing Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Product Type
- 16.4.3. Technology Platform
- 16.4.4. Sample Type
- 16.4.5. Mode of Purchase
- 16.4.6. Testing Complexity
- 16.4.7. Test Result Time
- 16.4.8. Connectivity
- 16.4.9. End-Use
- 16.5. Canada Point of Care Testing Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Product Type
- 16.5.3. Technology Platform
- 16.5.4. Sample Type
- 16.5.5. Mode of Purchase
- 16.5.6. Testing Complexity
- 16.5.7. Test Result Time
- 16.5.8. Connectivity
- 16.5.9. End-Use
- 16.6. Mexico Point of Care Testing Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Product Type
- 16.6.3. Technology Platform
- 16.6.4. Sample Type
- 16.6.5. Mode of Purchase
- 16.6.6. Testing Complexity
- 16.6.7. Test Result Time
- 16.6.8. Connectivity
- 16.6.9. End-Use
- 17. Europe Point of Care Testing Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Europe Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Product Type
- 17.3.2. Technology Platform
- 17.3.3. Sample Type
- 17.3.4. Mode of Purchase
- 17.3.5. Testing Complexity
- 17.3.6. Test Result Time
- 17.3.7. Connectivity
- 17.3.8. End-Use
- 17.3.9. Country
- 17.3.9.1. Germany
- 17.3.9.2. United Kingdom
- 17.3.9.3. France
- 17.3.9.4. Italy
- 17.3.9.5. Spain
- 17.3.9.6. Netherlands
- 17.3.9.7. Nordic Countries
- 17.3.9.8. Poland
- 17.3.9.9. Russia & CIS
- 17.3.9.10. Rest of Europe
- 17.4. Germany Point of Care Testing Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Product Type
- 17.4.3. Technology Platform
- 17.4.4. Sample Type
- 17.4.5. Mode of Purchase
- 17.4.6. Testing Complexity
- 17.4.7. Test Result Time
- 17.4.8. Connectivity
- 17.4.9. End-Use
- 17.5. United Kingdom Point of Care Testing Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Product Type
- 17.5.3. Technology Platform
- 17.5.4. Sample Type
- 17.5.5. Mode of Purchase
- 17.5.6. Testing Complexity
- 17.5.7. Test Result Time
- 17.5.8. Connectivity
- 17.5.9. End-Use
- 17.6. France Point of Care Testing Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Product Type
- 17.6.3. Technology Platform
- 17.6.4. Sample Type
- 17.6.5. Mode of Purchase
- 17.6.6. Testing Complexity
- 17.6.7. Test Result Time
- 17.6.8. Connectivity
- 17.6.9. End-Use
- 17.7. Italy Point of Care Testing Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Product Type
- 17.7.3. Technology Platform
- 17.7.4. Sample Type
- 17.7.5. Mode of Purchase
- 17.7.6. Testing Complexity
- 17.7.7. Test Result Time
- 17.7.8. Connectivity
- 17.7.9. End-Use
- 17.8. Spain Point of Care Testing Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Product Type
- 17.8.3. Technology Platform
- 17.8.4. Sample Type
- 17.8.5. Mode of Purchase
- 17.8.6. Testing Complexity
- 17.8.7. Test Result Time
- 17.8.8. Connectivity
- 17.8.9. End-Use
- 17.9. Netherlands Point of Care Testing Market
- 17.9.1. Country Segmental Analysis
- 17.9.2. Product Type
- 17.9.3. Technology Platform
- 17.9.4. Sample Type
- 17.9.5. Mode of Purchase
- 17.9.6. Testing Complexity
- 17.9.7. Test Result Time
- 17.9.8. Connectivity
- 17.9.9. End-Use
- 17.10. Nordic Countries Point of Care Testing Market
- 17.10.1. Country Segmental Analysis
- 17.10.2. Product Type
- 17.10.3. Technology Platform
- 17.10.4. Sample Type
- 17.10.5. Mode of Purchase
- 17.10.6. Testing Complexity
- 17.10.7. Test Result Time
- 17.10.8. Connectivity
- 17.10.9. End-Use
- 17.11. Poland Point of Care Testing Market
- 17.11.1. Country Segmental Analysis
- 17.11.2. Product Type
- 17.11.3. Technology Platform
- 17.11.4. Sample Type
- 17.11.5. Mode of Purchase
- 17.11.6. Testing Complexity
- 17.11.7. Test Result Time
- 17.11.8. Connectivity
- 17.11.9. End-Use
- 17.12. Russia & CIS Point of Care Testing Market
- 17.12.1. Country Segmental Analysis
- 17.12.2. Product Type
- 17.12.3. Technology Platform
- 17.12.4. Sample Type
- 17.12.5. Mode of Purchase
- 17.12.6. Testing Complexity
- 17.12.7. Test Result Time
- 17.12.8. Connectivity
- 17.12.9. End-Use
- 17.13. Rest of Europe Point of Care Testing Market
- 17.13.1. Country Segmental Analysis
- 17.13.2. Product Type
- 17.13.3. Technology Platform
- 17.13.4. Sample Type
- 17.13.5. Mode of Purchase
- 17.13.6. Testing Complexity
- 17.13.7. Test Result Time
- 17.13.8. Connectivity
- 17.13.9. End-Use
- 18. Asia Pacific Point of Care Testing Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Asia Pacific Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Product Type
- 18.3.2. Technology Platform
- 18.3.3. Sample Type
- 18.3.4. Mode of Purchase
- 18.3.5. Testing Complexity
- 18.3.6. Test Result Time
- 18.3.7. Connectivity
- 18.3.8. End-Use
- 18.3.9. Country
- 18.3.9.1. China
- 18.3.9.2. India
- 18.3.9.3. Japan
- 18.3.9.4. South Korea
- 18.3.9.5. Australia and New Zealand
- 18.3.9.6. Indonesia
- 18.3.9.7. Malaysia
- 18.3.9.8. Thailand
- 18.3.9.9. Vietnam
- 18.3.9.10. Rest of Asia Pacific
- 18.4. China Point of Care Testing Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Product Type
- 18.4.3. Technology Platform
- 18.4.4. Sample Type
- 18.4.5. Mode of Purchase
- 18.4.6. Testing Complexity
- 18.4.7. Test Result Time
- 18.4.8. Connectivity
- 18.4.9. End-Use
- 18.5. India Point of Care Testing Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Product Type
- 18.5.3. Technology Platform
- 18.5.4. Sample Type
- 18.5.5. Mode of Purchase
- 18.5.6. Testing Complexity
- 18.5.7. Test Result Time
- 18.5.8. Connectivity
- 18.5.9. End-Use
- 18.6. Japan Point of Care Testing Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Product Type
- 18.6.3. Technology Platform
- 18.6.4. Sample Type
- 18.6.5. Mode of Purchase
- 18.6.6. Testing Complexity
- 18.6.7. Test Result Time
- 18.6.8. Connectivity
- 18.6.9. End-Use
- 18.7. South Korea Point of Care Testing Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Product Type
- 18.7.3. Technology Platform
- 18.7.4. Sample Type
- 18.7.5. Mode of Purchase
- 18.7.6. Testing Complexity
- 18.7.7. Test Result Time
- 18.7.8. Connectivity
- 18.7.9. End-Use
- 18.8. Australia and New Zealand Point of Care Testing Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Product Type
- 18.8.3. Technology Platform
- 18.8.4. Sample Type
- 18.8.5. Mode of Purchase
- 18.8.6. Testing Complexity
- 18.8.7. Test Result Time
- 18.8.8. Connectivity
- 18.8.9. End-Use
- 18.9. Indonesia Point of Care Testing Market
- 18.9.1. Country Segmental Analysis
- 18.9.2. Product Type
- 18.9.3. Technology Platform
- 18.9.4. Sample Type
- 18.9.5. Mode of Purchase
- 18.9.6. Testing Complexity
- 18.9.7. Test Result Time
- 18.9.8. Connectivity
- 18.9.9. End-Use
- 18.10. Malaysia Point of Care Testing Market
- 18.10.1. Country Segmental Analysis
- 18.10.2. Product Type
- 18.10.3. Technology Platform
- 18.10.4. Sample Type
- 18.10.5. Mode of Purchase
- 18.10.6. Testing Complexity
- 18.10.7. Test Result Time
- 18.10.8. Connectivity
- 18.10.9. End-Use
- 18.11. Thailand Point of Care Testing Market
- 18.11.1. Country Segmental Analysis
- 18.11.2. Product Type
- 18.11.3. Technology Platform
- 18.11.4. Sample Type
- 18.11.5. Mode of Purchase
- 18.11.6. Testing Complexity
- 18.11.7. Test Result Time
- 18.11.8. Connectivity
- 18.11.9. End-Use
- 18.12. Vietnam Point of Care Testing Market
- 18.12.1. Country Segmental Analysis
- 18.12.2. Product Type
- 18.12.3. Technology Platform
- 18.12.4. Sample Type
- 18.12.5. Mode of Purchase
- 18.12.6. Testing Complexity
- 18.12.7. Test Result Time
- 18.12.8. Connectivity
- 18.12.9. End-Use
- 18.13. Rest of Asia Pacific Point of Care Testing Market
- 18.13.1. Country Segmental Analysis
- 18.13.2. Product Type
- 18.13.3. Technology Platform
- 18.13.4. Sample Type
- 18.13.5. Mode of Purchase
- 18.13.6. Testing Complexity
- 18.13.7. Test Result Time
- 18.13.8. Connectivity
- 18.13.9. End-Use
- 19. Middle East Point of Care Testing Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. Middle East Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Product Type
- 19.3.2. Technology Platform
- 19.3.3. Sample Type
- 19.3.4. Mode of Purchase
- 19.3.5. Testing Complexity
- 19.3.6. Test Result Time
- 19.3.7. Connectivity
- 19.3.8. End-Use
- 19.3.9. Country
- 19.3.9.1. Turkey
- 19.3.9.2. UAE
- 19.3.9.3. Saudi Arabia
- 19.3.9.4. Israel
- 19.3.9.5. Rest of Middle East
- 19.4. Turkey Point of Care Testing Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Product Type
- 19.4.3. Technology Platform
- 19.4.4. Sample Type
- 19.4.5. Mode of Purchase
- 19.4.6. Testing Complexity
- 19.4.7. Test Result Time
- 19.4.8. Connectivity
- 19.4.9. End-Use
- 19.5. UAE Point of Care Testing Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Product Type
- 19.5.3. Technology Platform
- 19.5.4. Sample Type
- 19.5.5. Mode of Purchase
- 19.5.6. Testing Complexity
- 19.5.7. Test Result Time
- 19.5.8. Connectivity
- 19.5.9. End-Use
- 19.6. Saudi Arabia Point of Care Testing Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Product Type
- 19.6.3. Technology Platform
- 19.6.4. Sample Type
- 19.6.5. Mode of Purchase
- 19.6.6. Testing Complexity
- 19.6.7. Test Result Time
- 19.6.8. Connectivity
- 19.6.9. End-Use
- 19.7. Israel Point of Care Testing Market
- 19.7.1. Country Segmental Analysis
- 19.7.2. Product Type
- 19.7.3. Technology Platform
- 19.7.4. Sample Type
- 19.7.5. Mode of Purchase
- 19.7.6. Testing Complexity
- 19.7.7. Test Result Time
- 19.7.8. Connectivity
- 19.7.9. End-Use Age Group
- 19.7.10. End-Use
- 19.8. Rest of Middle East Point of Care Testing Market
- 19.8.1. Country Segmental Analysis
- 19.8.2. Product Type
- 19.8.3. Technology Platform
- 19.8.4. Sample Type
- 19.8.5. Mode of Purchase
- 19.8.6. Testing Complexity
- 19.8.7. Test Result Time
- 19.8.8. Connectivity
- 19.8.9. End-Use
- 20. Africa Point of Care Testing Market Analysis
- 20.1. Key Segment Analysis
- 20.2. Regional Snapshot
- 20.3. Africa Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 20.3.1. Product Type
- 20.3.2. Technology Platform
- 20.3.3. Sample Type
- 20.3.4. Mode of Purchase
- 20.3.5. Testing Complexity
- 20.3.6. Test Result Time
- 20.3.7. Connectivity
- 20.3.8. End-Use
- 20.3.9. Country
- 20.3.9.1. South Africa
- 20.3.9.2. Egypt
- 20.3.9.3. Nigeria
- 20.3.9.4. Algeria
- 20.3.9.5. Rest of Africa
- 20.4. South Africa Point of Care Testing Market
- 20.4.1. Country Segmental Analysis
- 20.4.2. Product Type
- 20.4.3. Technology Platform
- 20.4.4. Sample Type
- 20.4.5. Mode of Purchase
- 20.4.6. Testing Complexity
- 20.4.7. Test Result Time
- 20.4.8. Connectivity
- 20.4.9. End-Use
- 20.5. Egypt Point of Care Testing Market
- 20.5.1. Country Segmental Analysis
- 20.5.2. Product Type
- 20.5.3. Technology Platform
- 20.5.4. Sample Type
- 20.5.5. Mode of Purchase
- 20.5.6. Testing Complexity
- 20.5.7. Test Result Time
- 20.5.8. Connectivity
- 20.5.9. End-Use
- 20.6. Nigeria Point of Care Testing Market
- 20.6.1. Country Segmental Analysis
- 20.6.2. Product Type
- 20.6.3. Technology Platform
- 20.6.4. Sample Type
- 20.6.5. Mode of Purchase
- 20.6.6. Testing Complexity
- 20.6.7. Test Result Time
- 20.6.8. Connectivity
- 20.6.9. End-Use
- 20.7. Algeria Point of Care Testing Market
- 20.7.1. Country Segmental Analysis
- 20.7.2. Product Type
- 20.7.3. Technology Platform
- 20.7.4. Sample Type
- 20.7.5. Mode of Purchase
- 20.7.6. Testing Complexity
- 20.7.7. Test Result Time
- 20.7.8. Connectivity
- 20.7.9. End-Use
- 20.8. Rest of Africa Point of Care Testing Market
- 20.8.1. Country Segmental Analysis
- 20.8.2. Product Type
- 20.8.3. Technology Platform
- 20.8.4. Sample Type
- 20.8.5. Mode of Purchase
- 20.8.6. Testing Complexity
- 20.8.7. Test Result Time
- 20.8.8. Connectivity
- 20.8.9. End-Use
- 21. South America Point of Care Testing Market Analysis
- 21.1. Key Segment Analysis
- 21.2. Regional Snapshot
- 21.3. South America Point of Care Testing Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 21.3.1. Product Type
- 21.3.2. Technology Platform
- 21.3.3. Sample Type
- 21.3.4. Mode of Purchase
- 21.3.5. Testing Complexity
- 21.3.6. Test Result Time
- 21.3.7. Connectivity
- 21.3.8. End-Use
- 21.3.9. Country
- 21.3.9.1. Brazil
- 21.3.9.2. Argentina
- 21.3.9.3. Rest of South America
- 21.4. Brazil Point of Care Testing Market
- 21.4.1. Country Segmental Analysis
- 21.4.2. Product Type
- 21.4.3. Technology Platform
- 21.4.4. Sample Type
- 21.4.5. Mode of Purchase
- 21.4.6. Testing Complexity
- 21.4.7. Test Result Time
- 21.4.8. Connectivity
- 21.4.9. End-Use
- 21.5. Argentina Point of Care Testing Market
- 21.5.1. Country Segmental Analysis
- 21.5.2. Product Type
- 21.5.3. Technology Platform
- 21.5.4. Sample Type
- 21.5.5. Mode of Purchase
- 21.5.6. Testing Complexity
- 21.5.7. Test Result Time
- 21.5.8. Connectivity
- 21.5.9. End-Use
- 21.6. Rest of South America Point of Care Testing Market
- 21.6.1. Country Segmental Analysis
- 21.6.2. Product Type
- 21.6.3. Technology Platform
- 21.6.4. Sample Type
- 21.6.5. Mode of Purchase
- 21.6.6. Testing Complexity
- 21.6.7. Test Result Time
- 21.6.8. Connectivity
- 21.6.9. End-Use
- 22. Key Players/ Company Profile
- 22.1. Abbott Laboratories
- 22.1.1. Company Details/ Overview
- 22.1.2. Company Financials
- 22.1.3. Key Customers and Competitors
- 22.1.4. Business/ Industry Portfolio
- 22.1.5. Product Portfolio/ Specification Details
- 22.1.6. Pricing Data
- 22.1.7. Strategic Overview
- 22.1.8. Recent Developments
- 22.2. Accriva Diagnostics
- 22.3. Becton, Dickinson and Company (BD)
- 22.4. bioMérieux
- 22.5. Chembio Diagnostics
- 22.6. Danaher Corporation
- 22.7. EKF Diagnostics
- 22.8. Instrumentation Laboratory
- 22.9. Johnson & Johnson
- 22.10. Mesa Biotech
- 22.11. Nipro Corporation
- 22.12. Nova Biomedical
- 22.13. Polymedco
- 22.14. PTS Diagnostics
- 22.15. Quidel Corporation
- 22.16. Roche Diagnostics
- 22.17. Sekisui Diagnostics
- 22.18. Siemens Healthineers
- 22.19. Sight Diagnostics
- 22.20. Trinity Biotech
- 22.21. Other Key Players
- 22.1. Abbott Laboratories
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
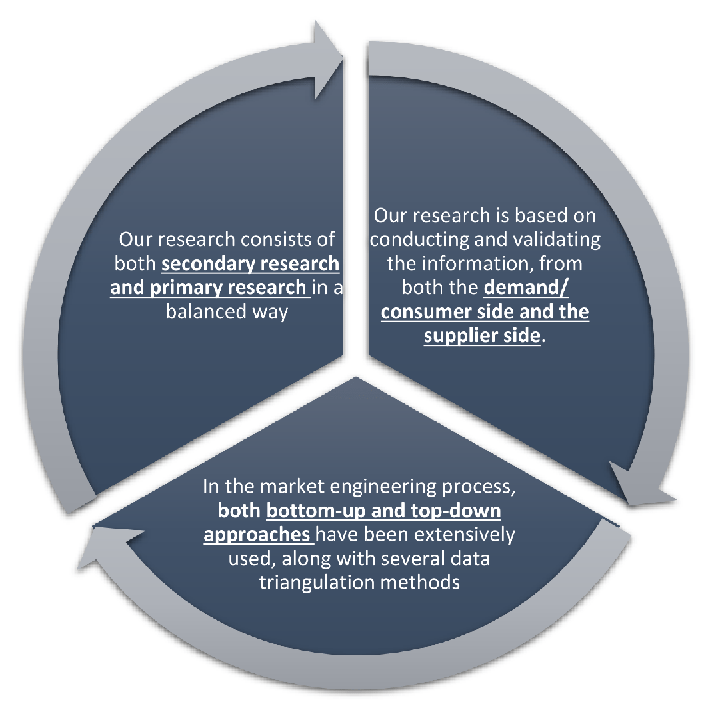
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
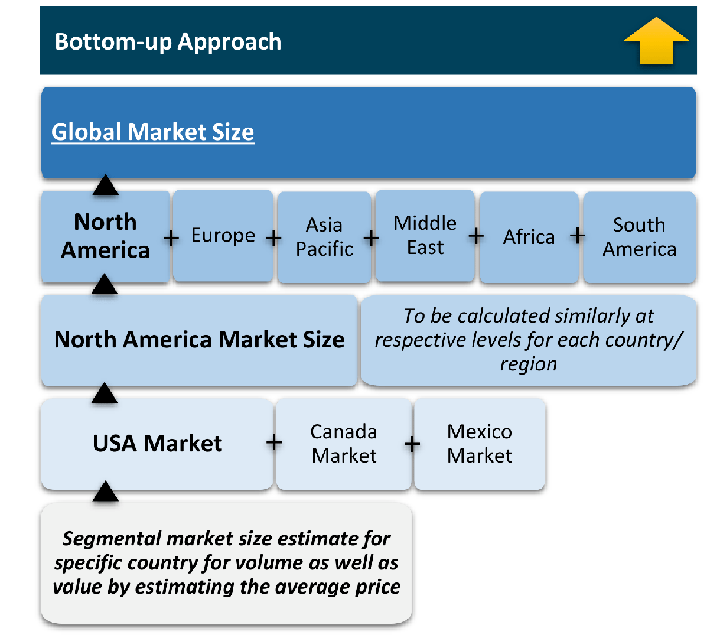
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation