Virtual Clinical Trials Market Size, Share & Trends Analysis Report by Trial Type (Fully Decentralized Trials, Hybrid Trials, Site-Based Trials with Virtual Components), Study Design, Phase, Component, Technology, Therapeutic Area, Patient Engagement Model, End User and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2025–2035
|
Market Structure & Evolution |
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
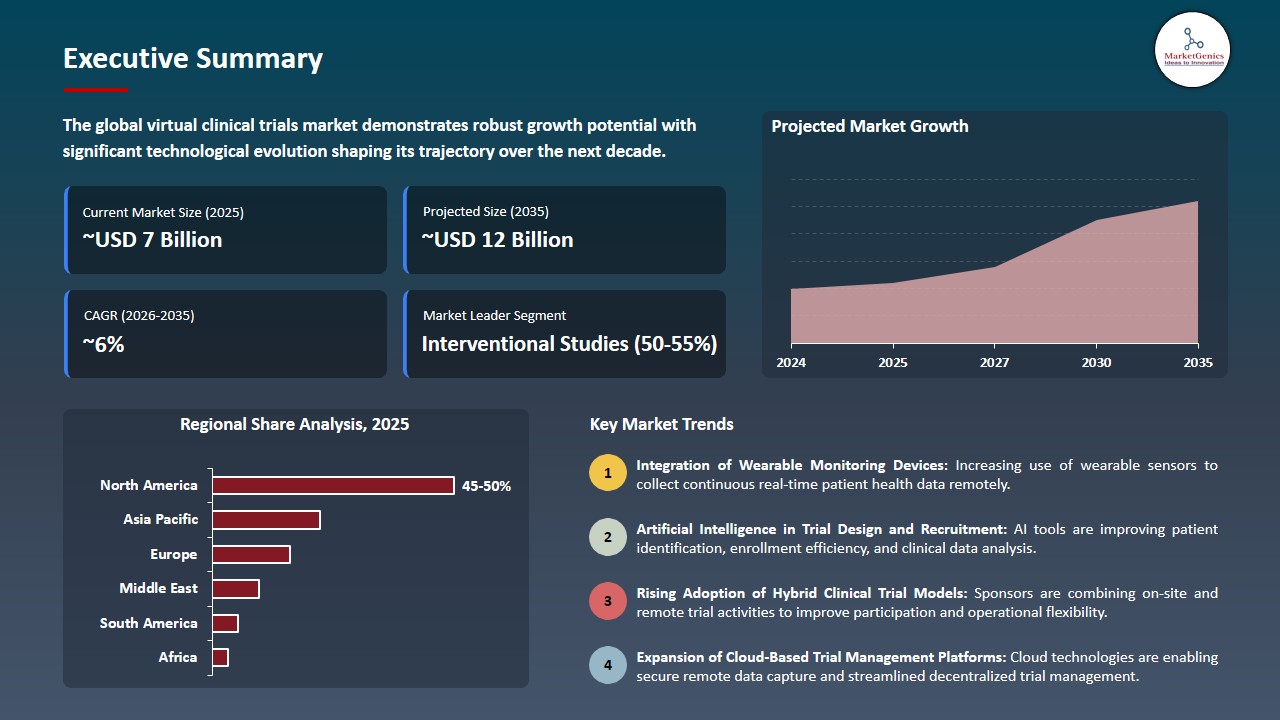
Virtual Clinical Trials Market Size, Share, and Growth
The global virtual clinical trials market is experiencing robust growth, with its estimated value of USD 6.6 billion in the year 2025 and USD 11.7 billion by 2035, registering a CAGR of 5.9% during the forecast period.
The partnership is expected to drive widespread implementation of our platform functions which is expected to assist life sciences companies to enhance patient access to clinical trials while they speed up their vital development work across the world according to Dr. Michelle Longmire who serves as both Co-founder and CEO of Medable.
The worldwide growth of the virtual clinical trials market results from decentralized trial platform adoption together with remote patient monitoring technology implementation which helps achieve better trial efficiency and greater patient access. For instance, in January 2024 Medable established a partnership with Google Cloud to deliver decentralized clinical trial functionality through their cloud-based analytics and artificial intelligence systems which help sponsors conduct remote patient data management and enhance trial processes.
Decentralized and hybrid models gain popularity because clinical researchers require better patient recruitment methods and faster trial execution times. Science 37 and Medidata Solutions joined forces in October 2023 to develop a system which connects decentralized trial technology with regular clinical research systems that enable remote patient visits and digital consent processes and continuous patient monitoring.
Pharmaceutical companies now adopt virtual trial models because regulatory agencies such as the U.S. Food and Drug Administration and the European Medicines Agency show growing acceptance of these methods. The market also has adjacent opportunities which include wearable health monitoring devices and telemedicine platforms and electronic patient-reported outcome systems and cloud-based clinical data management solutions.

Virtual Clinical Trials Market Dynamics and Trends
Driver: Rising Adoption of Decentralized and Hybrid Trial Models Accelerating Market Growth
-
The virtual clinical trials market experiences rapid expansion because pharmaceutical companies are implementing decentralized and hybrid trial models which help them achieve better patient recruitment and retention and operational efficiency. Trials can now access larger patient groups because digital consent platforms and remote monitoring tools and telehealth visits enable researchers to decrease their dependency on physical trial locations.
- The increasing complexity of clinical trials together with the need to recruit patients from worldwide locations pushes sponsors to adopt digital platforms which support remote participation and ongoing data collection. Pfizer established a partnership with Care Access in 2023 to enhance decentralized clinical research programs which allow more patients to participate in trials through local hybrid research models.
- The adoption of virtual trial technologies by pharmaceutical and biotechnology companies worldwide receives strong support because organizations require shorter clinical development times and better patient-centric research methods. All these factors are likely to continue to escalate the growth of the virtual clinical trials market.
Restraint: Data Privacy Concerns and Regulatory Complexity Limiting Adoption
-
The virtual clinical trial system shows strong development potential. The system faces obstacles which include maintaining patient data privacy and adhering to regulatory standards and managing data transfers between different countries. The digital platforms that handle patient-generated health data face risks to their data security measures together with the need for regulatory supervision.
- Companies must establish secure systems for protecting patient information and handling consent processes and storing data in order to meet both General Data Protection Regulation requirements and U.S. Food and Drug Administration agency requirements.
- Global decentralized trials face sponsors with two main obstacles which include the need to connect digital trial platforms to existing clinical systems and the need to achieve regulatory approval in different parts of the world. All these elements are expected to restrict the expansion of the virtual clinical trials market.
Opportunity: Expansion of Digital Health Infrastructure and Telemedicine Ecosystems
-
The digital health infrastructure and telemedicine platforms are expanding at a rapid pace which results in strong opportunities for virtual clinical trial adoption in areas that experience rising internet use and mobile health implementation. Researchers use remote healthcare delivery systems to access patients who live in remote or underserved locations.
- Pharmaceutical companies have obtained decentralized trial capabilities through their partnerships with growing technology providers. Oracle developed the Oracle Clinical One platform in 2022 by adding decentralized clinical trial features which enabled remote data collection and patient observation along with efficient trial operations.
- Digital health providers and wearable technology manufacturers together with telemedicine services is expected to benefit from these developments because they enable patient-centered research and help speed up drug development processes. And thus, is expected to create more opportunities in future for virtual clinical trials market.
Key Trend: Integration of Wearables, Real-World Data, and Artificial Intelligence in Clinical Research
-
The virtual clinical trials market currently undergoes a significant transformation because organizations are using wearable health devices together with real-world data collection techniques and artificial intelligence analytics to enhance both patient monitoring processes and trial data accuracy. Wearable devices combined with mobile health applications provide continuous patient health metric monitoring which occurs beyond the boundaries of standard clinical environments.
- The growing adoption of artificial intelligence tools has enabled researchers to find suitable trial participants while they process massive amounts of patient data and design more effective clinical studies. IQVIA introduced new decentralized trial capabilities in 2023 when it launched its IQVIA Decentralized Trials solutions to enable remote patient monitoring and advanced analytics for patient-centered research studies.
- The combination of digital therapeutics with wearable sensors and real-world evidence platforms has created a new research method which is expected to boost research productivity while involving more patients in upcoming years. Therefore, is expected to influence significant trends in the virtual clinical trials market.
Virtual Clinical Trials Market Analysis and Segmental Data
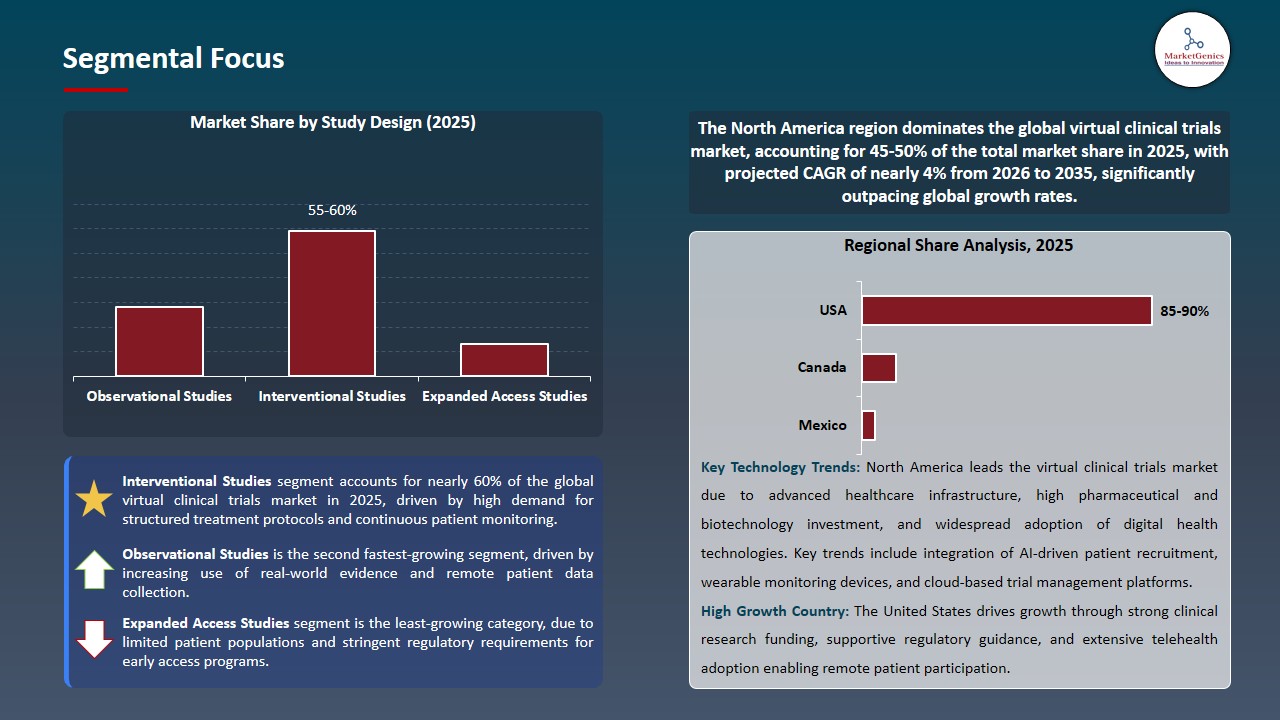
Interventional Studies Dominates Global Virtual Clinical Trials Market amid Rising Adoption of Decentralized Drug Trials
-
The Interventional Studies segment controls the virtual clinical trials market because drug and biologic studies require research procedures that include structured treatment protocols along with continuous patient observation and authorized assessment methods.
- Pharmaceutical and biotechnology companies are adopting decentralized tools which include remote patient monitoring and digital consent and telehealth visits to enhance their participant recruitment and retention efforts during interventional trials. The clinical data needs of these trials require digital platforms to enable instant data collection and regulatory requirements fulfillment.
- Notably, Parexel expanded its decentralized clinical trial capabilities through a partnership with Palantir Technologies which enables the company to enhance digital trial execution and data integration for its worldwide interventional studies marking interventional studies as the leading segment in virtual clinical trials market.
North America Dominates Virtual Clinical Trials Market amid Strong Digital Health Infrastructure and High Clinical Research Investment
-
The virtual clinical trials market in North America because the region has developed advanced digital health systems and it has many pharmaceutical and biotechnology companies and most of its people use telehealth and electronic health record systems to conduct decentralized research.
- The region enjoys advantages from its regulatory framework and the United States invests heavily in clinical research which drives worldwide progress in digital clinical trial technology.
- The U.S. Food and Drug Administration published updated guidance in May 2023 which recommends using decentralized clinical trials for drugs and biologics and medical devices to allow remote patient participation while providing flexible trial procedures, is expected to continue to strengthen virtual clinical trials market in North America.
Virtual Clinical Trials Market Ecosystem
The virtual clinical trials market is highly consolidated with organizations operating alongside digital health technology providers which create moderate market fragmentation. The integrated decentralized trial solutions of IQVIA and ICON plc establish them as Tier-1 market leaders while Medable and Science 37 operate specialized digital platforms as Tier-2 and Tier-3 companies.
The vital components of the value chain operate through digital trial technology platforms and clinical research organization services which support patient recruitment and trial management activities. ICON plc developed new decentralized trial solutions in 2024 through its remote patient monitoring technology.

Recent Development and Strategic Overview:
-
In August 2025, Veeva established a strategic partnership with Amgen which is a worldwide biotechnology enterprise to utilize the Veeva Clinical Platform for developing new clinical trial methods and discovering operational improvements in their trial procedures. The partnership demonstrates how Veeva continues to assist clinical operations which use digital technologies to conduct hybrid and decentralized research studies.
- In March 2024, Medidata Solutions launched new decentralized trial features for its Medidata Platform which enables clinical researchers to use wearable device data and remote consent and virtual patient visits for more efficient execution of decentralized clinical studies and better access to trial data in real time.
Report Scope
|
Attribute |
Detail |
|
Market Size in 2025 |
USD 6.6 Bn |
|
Market Forecast Value in 2035 |
USD 11.7 Bn |
|
Growth Rate (CAGR) |
5.9% |
|
Forecast Period |
2025 – 2035 |
|
Historical Data Available for |
2020 – 2024 |
|
Market Size Units |
USD Billion for Value |
|
Report Format |
Electronic (PDF) + Excel |
|
Regions and Countries Covered |
|||||
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Virtual Clinical Trials Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Virtual Clinical Trials Market, By Trial Type |
|
|
Virtual Clinical Trials Market, By Study Design |
|
|
Virtual Clinical Trials Market, By Phase |
|
|
Virtual Clinical Trials Market, By Component |
|
|
Virtual Clinical Trials Market, By Technology |
|
|
Virtual Clinical Trials Market, By Therapeutic Area |
|
|
Virtual Clinical Trials Market, By Patient Engagement Model |
|
|
Virtual Clinical Trials Market, By Data Collection Method |
|
|
Virtual Clinical Trials Market, By End User |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Natures
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Natures
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Virtual Clinical Trials Market Outlook
- 2.1.1. Virtual Clinical Trials Market Size (Value - US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Virtual Clinical Trials Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Industry Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Growing adoption of decentralized and remote healthcare solutions.
- 4.1.1.2. Increased need for faster and cost-effective clinical trial processes.
- 4.1.1.3. Rising prevalence of chronic diseases boosting demand for patient-centric trials.
- 4.1.2. Restraints
- 4.1.2.1. Data privacy and cybersecurity concerns in handling patient information.
- 4.1.2.2. Limited technological infrastructure in emerging markets restricting adoption.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.5. Cost Structure Analysis
- 4.6. Porter’s Five Forces Analysis
- 4.7. PESTEL Analysis
- 4.8. Global Virtual Clinical Trials Market Demand
- 4.8.1. Historical Market Size – Value (US$ Bn), 2020-2024
- 4.8.2. Current and Future Market Size – Value (US$ Bn), 2026–2035
- 4.8.2.1. Y-o-Y Growth Trends
- 4.8.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Virtual Clinical Trials Market Analysis, by Trial Type
- 6.1. Key Segment Analysis
- 6.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Trial Type, 2021-2035
- 6.2.1. Fully Decentralized Trials
- 6.2.2. Hybrid Trials
- 6.2.3. Site-Based Trials with Virtual Components
- 7. Global Virtual Clinical Trials Market Analysis, by Study Design
- 7.1. Key Segment Analysis
- 7.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Study Design, 2021-2035
- 7.2.1. Interventional Studies
- 7.2.2. Observational Studies
- 7.2.3. Expanded Access Studies
- 8. Global Virtual Clinical Trials Market Analysis, by Phase
- 8.1. Key Segment Analysis
- 8.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Phase, 2021-2035
- 8.2.1. Phase I
- 8.2.2. Phase II
- 8.2.3. Phase III
- 8.2.4. Phase IV
- 9. Global Virtual Clinical Trials Market Analysis, by Component
- 9.1. Key Segment Analysis
- 9.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Component, 2021-2035
- 9.2.1. Software Platforms
- 9.2.1.1. Clinical Trial Management Systems (CTMS)
- 9.2.1.2. Electronic Data Capture (EDC) Systems
- 9.2.1.3. Electronic Clinical Outcome Assessment (eCOA) Platforms
- 9.2.1.4. Electronic Patient-Reported Outcome (ePRO) Systems
- 9.2.1.5. eConsent Platforms
- 9.2.1.6. Telemedicine and Virtual Visit Platforms
- 9.2.1.7. Remote Patient Monitoring (RPM) Software
- 9.2.1.8. Patient Recruitment and Engagement Platforms
- 9.2.1.9. Randomization and Trial Supply Management (RTSM) Systems
- 9.2.1.10. Clinical Data Management Systems (CDMS)
- 9.2.1.11. Digital Biomarker Analytics Platforms
- 9.2.1.12. AI-Based Clinical Trial Analytics Platforms
- 9.2.1.13. Data Integration and Interoperability Platforms
- 9.2.1.14. Regulatory Compliance and Documentation Software
- 9.2.1.15. Others
- 9.2.2. Services
- 9.2.2.1. Decentralized Clinical Trial (DCT) Design and Consulting
- 9.2.2.2. Virtual Clinical Trial Implementation Services
- 9.2.2.3. Patient Recruitment and Retention Services
- 9.2.2.4. Remote Monitoring and Site Management Services
- 9.2.2.5. Telehealth and Virtual Visit Support Services
- 9.2.2.6. Home Healthcare and Mobile Nursing Services
- 9.2.2.7. Data Management and Biostatistics Services
- 9.2.2.8. Regulatory Affairs and Compliance Services
- 9.2.2.9. Logistics and Drug Supply Management
- 9.2.2.10. Technology Integration and Platform Support
- 9.2.2.11. Training and Technical Support Services
- 9.2.2.12. Others
- 9.2.3. Hardware and Connected Devices
- 9.2.3.1. Wearable Health Monitoring Devices
- 9.2.3.2. Biosensors and Digital Biomarker Devices
- 9.2.3.3. Smart Medical Devices (Connected Devices)
- 9.2.3.4. Remote Diagnostic Devices
- 9.2.3.5. Mobile Devices (Smartphones and Tablets)
- 9.2.3.6. Patient Monitoring Equipment
- 9.2.3.7. Home-Based Clinical Trial Kits
- 9.2.3.8. Smart Medication Adherence Devices
- 9.2.3.9. Telehealth Communication Devices
- 9.2.3.10. Portable Diagnostic and Imaging Devices
- 9.2.3.11. Others
- 9.2.1. Software Platforms
- 10. Global Virtual Clinical Trials Market Analysis, by Technology
- 10.1. Key Segment Analysis
- 10.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Technology, 2021-2035
- 10.2.1. Telemedicine Platforms
- 10.2.2. Electronic Data Capture (EDC) Systems
- 10.2.3. eConsent Platforms
- 10.2.4. Mobile Health (mHealth) Applications
- 10.2.5. Wearable Devices and Biosensors
- 10.2.6. Remote Patient Monitoring Platforms
- 10.2.7. Clinical Trial Management Systems (CTMS)
- 10.2.8. eClinical Platforms
- 10.2.9. Others
- 11. Global Virtual Clinical Trials Market Analysis, by Therapeutic Area
- 11.1. Key Segment Analysis
- 11.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Therapeutic Area, 2021-2035
- 11.2.1. Oncology
- 11.2.2. Cardiovascular Diseases
- 11.2.3. Central Nervous System (CNS) Disorders
- 11.2.4. Infectious Diseases
- 11.2.5. Metabolic and Endocrine Disorders
- 11.2.6. Respiratory Diseases
- 11.2.7. Immunology and Autoimmune Diseases
- 11.2.8. Rare Diseases
- 11.2.9. Others
- 12. Global Virtual Clinical Trials Market Analysis, by Patient Engagement Model
- 12.1. Key Segment Analysis
- 12.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Patient Engagement Model, 2021-2035
- 12.2.1. Direct-to-Patient Trials
- 12.2.2. Home Healthcare Supported Trials
- 12.2.3. Hybrid Patient Engagement Models
- 13. Global Virtual Clinical Trials Market Analysis, by Data Collection Method
- 13.1. Key Segment Analysis
- 13.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Data Collection Method, 2021-2035
- 13.2.1. Electronic Patient-Reported Outcomes (ePRO)
- 13.2.2. Sensor and Wearable-Based Monitoring
- 13.2.3. Mobile App-Based Data Collection
- 13.2.4. Teleconsultation and Virtual Visits
- 13.2.5. Others
- 14. Global Virtual Clinical Trials Market Analysis, by End User
- 14.1. Key Segment Analysis
- 14.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by End User, 2021-2035
- 14.2.1. Pharmaceutical Companies
- 14.2.2. Biotechnology Companies
- 14.2.3. Contract Research Organizations (CROs)
- 14.2.4. Medical Device Companies
- 14.2.5. Academic and Research Institutes
- 14.2.6. Others
- 15. Global Virtual Clinical Trials Market Analysis and Forecasts, by Region
- 15.1. Key Findings
- 15.2. Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 15.2.1. North America
- 15.2.2. Europe
- 15.2.3. Asia Pacific
- 15.2.4. Middle East
- 15.2.5. Africa
- 15.2.6. South America
- 16. North America Virtual Clinical Trials Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. North America Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Trial Type
- 16.3.2. Study Design
- 16.3.3. Phase
- 16.3.4. Component
- 16.3.5. Technology
- 16.3.6. Therapeutic Area
- 16.3.7. Patient Engagement Model
- 16.3.8. Data Collection Method
- 16.3.9. End User
- 16.3.10. Country
- 16.3.10.1. USA
- 16.3.10.2. Canada
- 16.3.10.3. Mexico
- 16.4. USA Virtual Clinical Trials Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Trial Type
- 16.4.3. Study Design
- 16.4.4. Phase
- 16.4.5. Component
- 16.4.6. Technology
- 16.4.7. Therapeutic Area
- 16.4.8. Patient Engagement Model
- 16.4.9. Data Collection Method
- 16.4.10. End User
- 16.5. Canada Virtual Clinical Trials Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Trial Type
- 16.5.3. Study Design
- 16.5.4. Phase
- 16.5.5. Component
- 16.5.6. Technology
- 16.5.7. Therapeutic Area
- 16.5.8. Patient Engagement Model
- 16.5.9. Data Collection Method
- 16.5.10. End User
- 16.6. Mexico Virtual Clinical Trials Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Trial Type
- 16.6.3. Study Design
- 16.6.4. Phase
- 16.6.5. Component
- 16.6.6. Technology
- 16.6.7. Therapeutic Area
- 16.6.8. Patient Engagement Model
- 16.6.9. Data Collection Method
- 16.6.10. End User
- 17. Europe Virtual Clinical Trials Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Europe Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Trial Type
- 17.3.2. Study Design
- 17.3.3. Phase
- 17.3.4. Component
- 17.3.5. Technology
- 17.3.6. Therapeutic Area
- 17.3.7. Patient Engagement Model
- 17.3.8. Data Collection Method
- 17.3.9. End User
- 17.3.10. Country
- 17.3.10.1. Germany
- 17.3.10.2. United Kingdom
- 17.3.10.3. France
- 17.3.10.4. Italy
- 17.3.10.5. Spain
- 17.3.10.6. Netherlands
- 17.3.10.7. Nordic Countries
- 17.3.10.8. Poland
- 17.3.10.9. Russia & CIS
- 17.3.10.10. Rest of Europe
- 17.4. Germany Virtual Clinical Trials Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Trial Type
- 17.4.3. Study Design
- 17.4.4. Phase
- 17.4.5. Component
- 17.4.6. Technology
- 17.4.7. Therapeutic Area
- 17.4.8. Patient Engagement Model
- 17.4.9. Data Collection Method
- 17.4.10. End User
- 17.5. United Kingdom Virtual Clinical Trials Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Trial Type
- 17.5.3. Study Design
- 17.5.4. Phase
- 17.5.5. Component
- 17.5.6. Technology
- 17.5.7. Therapeutic Area
- 17.5.8. Patient Engagement Model
- 17.5.9. Data Collection Method
- 17.5.10. End User
- 17.6. France Virtual Clinical Trials Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Trial Type
- 17.6.3. Study Design
- 17.6.4. Phase
- 17.6.5. Component
- 17.6.6. Technology
- 17.6.7. Therapeutic Area
- 17.6.8. Patient Engagement Model
- 17.6.9. Data Collection Method
- 17.6.10. End User
- 17.7. Italy Virtual Clinical Trials Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Trial Type
- 17.7.3. Study Design
- 17.7.4. Phase
- 17.7.5. Component
- 17.7.6. Technology
- 17.7.7. Therapeutic Area
- 17.7.8. Patient Engagement Model
- 17.7.9. Data Collection Method
- 17.7.10. End User
- 17.8. Spain Virtual Clinical Trials Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Trial Type
- 17.8.3. Study Design
- 17.8.4. Phase
- 17.8.5. Component
- 17.8.6. Technology
- 17.8.7. Therapeutic Area
- 17.8.8. Patient Engagement Model
- 17.8.9. Data Collection Method
- 17.8.10. End User
- 17.9. Netherlands Virtual Clinical Trials Market
- 17.9.1. Country Segmental Analysis
- 17.9.2. Trial Type
- 17.9.3. Study Design
- 17.9.4. Phase
- 17.9.5. Component
- 17.9.6. Technology
- 17.9.7. Therapeutic Area
- 17.9.8. Patient Engagement Model
- 17.9.9. Data Collection Method
- 17.9.10. End User
- 17.10. Nordic Countries Virtual Clinical Trials Market
- 17.10.1. Country Segmental Analysis
- 17.10.2. Trial Type
- 17.10.3. Study Design
- 17.10.4. Phase
- 17.10.5. Component
- 17.10.6. Technology
- 17.10.7. Therapeutic Area
- 17.10.8. Patient Engagement Model
- 17.10.9. Data Collection Method
- 17.10.10. End User
- 17.11. Poland Virtual Clinical Trials Market
- 17.11.1. Country Segmental Analysis
- 17.11.2. Trial Type
- 17.11.3. Study Design
- 17.11.4. Phase
- 17.11.5. Component
- 17.11.6. Technology
- 17.11.7. Therapeutic Area
- 17.11.8. Patient Engagement Model
- 17.11.9. Data Collection Method
- 17.11.10. End User
- 17.12. Russia & CIS Virtual Clinical Trials Market
- 17.12.1. Country Segmental Analysis
- 17.12.2. Trial Type
- 17.12.3. Study Design
- 17.12.4. Phase
- 17.12.5. Component
- 17.12.6. Technology
- 17.12.7. Therapeutic Area
- 17.12.8. Patient Engagement Model
- 17.12.9. Data Collection Method
- 17.12.10. End User
- 17.13. Rest of Europe Virtual Clinical Trials Market
- 17.13.1. Country Segmental Analysis
- 17.13.2. Trial Type
- 17.13.3. Study Design
- 17.13.4. Phase
- 17.13.5. Component
- 17.13.6. Technology
- 17.13.7. Therapeutic Area
- 17.13.8. Patient Engagement Model
- 17.13.9. Data Collection Method
- 17.13.10. End User
- 18. Asia Pacific Virtual Clinical Trials Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Asia Pacific Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Trial Type
- 18.3.2. Study Design
- 18.3.3. Phase
- 18.3.4. Component
- 18.3.5. Technology
- 18.3.6. Therapeutic Area
- 18.3.7. Patient Engagement Model
- 18.3.8. Data Collection Method
- 18.3.9. End User
- 18.3.10. Country
- 18.3.10.1. China
- 18.3.10.2. India
- 18.3.10.3. Japan
- 18.3.10.4. South Korea
- 18.3.10.5. Australia and New Zealand
- 18.3.10.6. Indonesia
- 18.3.10.7. Malaysia
- 18.3.10.8. Thailand
- 18.3.10.9. Vietnam
- 18.3.10.10. Rest of Asia Pacific
- 18.4. China Virtual Clinical Trials Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Trial Type
- 18.4.3. Study Design
- 18.4.4. Phase
- 18.4.5. Component
- 18.4.6. Technology
- 18.4.7. Therapeutic Area
- 18.4.8. Patient Engagement Model
- 18.4.9. Data Collection Method
- 18.4.10. End User
- 18.5. India Virtual Clinical Trials Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Trial Type
- 18.5.3. Study Design
- 18.5.4. Phase
- 18.5.5. Component
- 18.5.6. Technology
- 18.5.7. Therapeutic Area
- 18.5.8. Patient Engagement Model
- 18.5.9. Data Collection Method
- 18.5.10. End User
- 18.6. Japan Virtual Clinical Trials Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Trial Type
- 18.6.3. Study Design
- 18.6.4. Phase
- 18.6.5. Component
- 18.6.6. Technology
- 18.6.7. Therapeutic Area
- 18.6.8. Patient Engagement Model
- 18.6.9. Data Collection Method
- 18.6.10. End User
- 18.7. South Korea Virtual Clinical Trials Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Trial Type
- 18.7.3. Study Design
- 18.7.4. Phase
- 18.7.5. Component
- 18.7.6. Technology
- 18.7.7. Therapeutic Area
- 18.7.8. Patient Engagement Model
- 18.7.9. Data Collection Method
- 18.7.10. End User
- 18.8. Australia and New Zealand Virtual Clinical Trials Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Trial Type
- 18.8.3. Study Design
- 18.8.4. Phase
- 18.8.5. Component
- 18.8.6. Technology
- 18.8.7. Therapeutic Area
- 18.8.8. Patient Engagement Model
- 18.8.9. Data Collection Method
- 18.8.10. End User
- 18.9. Indonesia Virtual Clinical Trials Market
- 18.9.1. Country Segmental Analysis
- 18.9.2. Trial Type
- 18.9.3. Study Design
- 18.9.4. Phase
- 18.9.5. Component
- 18.9.6. Technology
- 18.9.7. Therapeutic Area
- 18.9.8. Patient Engagement Model
- 18.9.9. Data Collection Method
- 18.9.10. End User
- 18.10. Malaysia Virtual Clinical Trials Market
- 18.10.1. Country Segmental Analysis
- 18.10.2. Trial Type
- 18.10.3. Study Design
- 18.10.4. Phase
- 18.10.5. Component
- 18.10.6. Technology
- 18.10.7. Therapeutic Area
- 18.10.8. Patient Engagement Model
- 18.10.9. Data Collection Method
- 18.10.10. End User
- 18.11. Thailand Virtual Clinical Trials Market
- 18.11.1. Country Segmental Analysis
- 18.11.2. Trial Type
- 18.11.3. Study Design
- 18.11.4. Phase
- 18.11.5. Component
- 18.11.6. Technology
- 18.11.7. Therapeutic Area
- 18.11.8. Patient Engagement Model
- 18.11.9. Data Collection Method
- 18.11.10. End User
- 18.12. Vietnam Virtual Clinical Trials Market
- 18.12.1. Country Segmental Analysis
- 18.12.2. Trial Type
- 18.12.3. Study Design
- 18.12.4. Phase
- 18.12.5. Component
- 18.12.6. Technology
- 18.12.7. Therapeutic Area
- 18.12.8. Patient Engagement Model
- 18.12.9. Data Collection Method
- 18.12.10. End User
- 18.13. Rest of Asia Pacific Virtual Clinical Trials Market
- 18.13.1. Country Segmental Analysis
- 18.13.2. Trial Type
- 18.13.3. Study Design
- 18.13.4. Phase
- 18.13.5. Component
- 18.13.6. Technology
- 18.13.7. Therapeutic Area
- 18.13.8. Patient Engagement Model
- 18.13.9. Data Collection Method
- 18.13.10. End User
- 19. Middle East Virtual Clinical Trials Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. Middle East Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Trial Type
- 19.3.2. Study Design
- 19.3.3. Phase
- 19.3.4. Component
- 19.3.5. Technology
- 19.3.6. Therapeutic Area
- 19.3.7. Patient Engagement Model
- 19.3.8. Data Collection Method
- 19.3.9. End User
- 19.3.10. Country
- 19.3.10.1. Turkey
- 19.3.10.2. UAE
- 19.3.10.3. Saudi Arabia
- 19.3.10.4. Israel
- 19.3.10.5. Rest of Middle East
- 19.4. Turkey Virtual Clinical Trials Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Trial Type
- 19.4.3. Study Design
- 19.4.4. Phase
- 19.4.5. Component
- 19.4.6. Technology
- 19.4.7. Therapeutic Area
- 19.4.8. Patient Engagement Model
- 19.4.9. Data Collection Method
- 19.4.10. End User
- 19.5. UAE Virtual Clinical Trials Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Trial Type
- 19.5.3. Study Design
- 19.5.4. Phase
- 19.5.5. Component
- 19.5.6. Technology
- 19.5.7. Therapeutic Area
- 19.5.8. Patient Engagement Model
- 19.5.9. Data Collection Method
- 19.5.10. End User
- 19.6. Saudi Arabia Virtual Clinical Trials Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Trial Type
- 19.6.3. Study Design
- 19.6.4. Phase
- 19.6.5. Component
- 19.6.6. Technology
- 19.6.7. Therapeutic Area
- 19.6.8. Patient Engagement Model
- 19.6.9. Data Collection Method
- 19.6.10. End User
- 19.7. Israel Virtual Clinical Trials Market
- 19.7.1. Country Segmental Analysis
- 19.7.2. Trial Type
- 19.7.3. Study Design
- 19.7.4. Phase
- 19.7.5. Component
- 19.7.6. Technology
- 19.7.7. Therapeutic Area
- 19.7.8. Patient Engagement Model
- 19.7.9. Data Collection Method
- 19.7.10. End User
- 19.8. Rest of Middle East Virtual Clinical Trials Market
- 19.8.1. Country Segmental Analysis
- 19.8.2. Trial Type
- 19.8.3. Study Design
- 19.8.4. Phase
- 19.8.5. Component
- 19.8.6. Technology
- 19.8.7. Therapeutic Area
- 19.8.8. Patient Engagement Model
- 19.8.9. Data Collection Method
- 19.8.10. End User
- 20. Africa Virtual Clinical Trials Market Analysis
- 20.1. Key Segment Analysis
- 20.2. Regional Snapshot
- 20.3. Africa Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 20.3.1. Trial Type
- 20.3.2. Study Design
- 20.3.3. Phase
- 20.3.4. Component
- 20.3.5. Technology
- 20.3.6. Therapeutic Area
- 20.3.7. Patient Engagement Model
- 20.3.8. Data Collection Method
- 20.3.9. End User
- 20.3.10. Country
- 20.3.10.1. South Africa
- 20.3.10.2. Egypt
- 20.3.10.3. Nigeria
- 20.3.10.4. Algeria
- 20.3.10.5. Rest of Africa
- 20.4. South Africa Virtual Clinical Trials Market
- 20.4.1. Country Segmental Analysis
- 20.4.2. Trial Type
- 20.4.3. Study Design
- 20.4.4. Phase
- 20.4.5. Component
- 20.4.6. Technology
- 20.4.7. Therapeutic Area
- 20.4.8. Patient Engagement Model
- 20.4.9. Data Collection Method
- 20.4.10. End User
- 20.5. Egypt Virtual Clinical Trials Market
- 20.5.1. Country Segmental Analysis
- 20.5.2. Trial Type
- 20.5.3. Study Design
- 20.5.4. Phase
- 20.5.5. Component
- 20.5.6. Technology
- 20.5.7. Therapeutic Area
- 20.5.8. Patient Engagement Model
- 20.5.9. Data Collection Method
- 20.5.10. End User
- 20.6. Nigeria Virtual Clinical Trials Market
- 20.6.1. Country Segmental Analysis
- 20.6.2. Trial Type
- 20.6.3. Study Design
- 20.6.4. Phase
- 20.6.5. Component
- 20.6.6. Technology
- 20.6.7. Therapeutic Area
- 20.6.8. Patient Engagement Model
- 20.6.9. Data Collection Method
- 20.6.10. End User
- 20.7. Algeria Virtual Clinical Trials Market
- 20.7.1. Country Segmental Analysis
- 20.7.2. Trial Type
- 20.7.3. Study Design
- 20.7.4. Phase
- 20.7.5. Component
- 20.7.6. Technology
- 20.7.7. Therapeutic Area
- 20.7.8. Patient Engagement Model
- 20.7.9. Data Collection Method
- 20.7.10. End User
- 20.8. Rest of Africa Virtual Clinical Trials Market
- 20.8.1. Country Segmental Analysis
- 20.8.2. Trial Type
- 20.8.3. Study Design
- 20.8.4. Phase
- 20.8.5. Component
- 20.8.6. Technology
- 20.8.7. Therapeutic Area
- 20.8.8. Patient Engagement Model
- 20.8.9. Data Collection Method
- 20.8.10. End User
- 21. South America Virtual Clinical Trials Market Analysis
- 21.1. Key Segment Analysis
- 21.2. Regional Snapshot
- 21.3. South America Virtual Clinical Trials Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 21.3.1. Trial Type
- 21.3.2. Study Design
- 21.3.3. Phase
- 21.3.4. Component
- 21.3.5. Technology
- 21.3.6. Therapeutic Area
- 21.3.7. Patient Engagement Model
- 21.3.8. Data Collection Method
- 21.3.9. End User
- 21.3.10. Country
- 21.3.10.1. Brazil
- 21.3.10.2. Argentina
- 21.3.10.3. Rest of South America
- 21.4. Brazil Virtual Clinical Trials Market
- 21.4.1. Country Segmental Analysis
- 21.4.2. Trial Type
- 21.4.3. Study Design
- 21.4.4. Phase
- 21.4.5. Component
- 21.4.6. Technology
- 21.4.7. Therapeutic Area
- 21.4.8. Patient Engagement Model
- 21.4.9. Data Collection Method
- 21.4.10. End User
- 21.5. Argentina Virtual Clinical Trials Market
- 21.5.1. Country Segmental Analysis
- 21.5.2. Trial Type
- 21.5.3. Study Design
- 21.5.4. Phase
- 21.5.5. Component
- 21.5.6. Technology
- 21.5.7. Therapeutic Area
- 21.5.8. Patient Engagement Model
- 21.5.9. Data Collection Method
- 21.5.10. End User
- 21.6. Rest of South America Virtual Clinical Trials Market
- 21.6.1. Country Segmental Analysis
- 21.6.2. Trial Type
- 21.6.3. Study Design
- 21.6.4. Trial Type
- 21.6.5. Component
- 21.6.6. Crop Type
- 21.6.7. Therapeutic Area
- 21.6.8. Patient Engagement Model
- 21.6.9. End User
- 22. Key Players/ Company Profile
- 22.1. Castor Research Inc.
- 22.1.1. Company Details/ Overview
- 22.1.2. Company Financials
- 22.1.3. Key Customers and Competitors
- 22.1.4. Business/ Industry Portfolio
- 22.1.5. Product Portfolio/ Specification Details
- 22.1.6. Pricing Data
- 22.1.7. Strategic Overview
- 22.1.8. Recent Developments
- 22.2. Clario (ERT Clinical)
- 22.3. Clinical Ink, Inc.
- 22.4. CRF Health, Inc.
- 22.5. Curebase, Inc.
- 22.6. Dassault Systèmes SE (Medidata Solutions)
- 22.7. ICON plc
- 22.8. IQVIA Holdings Inc.
- 22.9. Labcorp Holdings Inc.
- 22.10. Lightship, Inc.
- 22.11. Medable, Inc.
- 22.12. ObvioHealth, Inc.
- 22.13. Oracle Corporation
- 22.14. Parexel International Corporation
- 22.15. Science 37, Inc.
- 22.16. Signant Health Holding Corp.
- 22.17. Syneos Health, Inc.
- 22.18. Thermo Fisher Scientific Inc. (PPD, Inc.)
- 22.19. THREAD Research, Inc.
- 22.20. Veeva Systems Inc.
- 22.21. Other Key Players
- 22.1. Castor Research Inc.
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
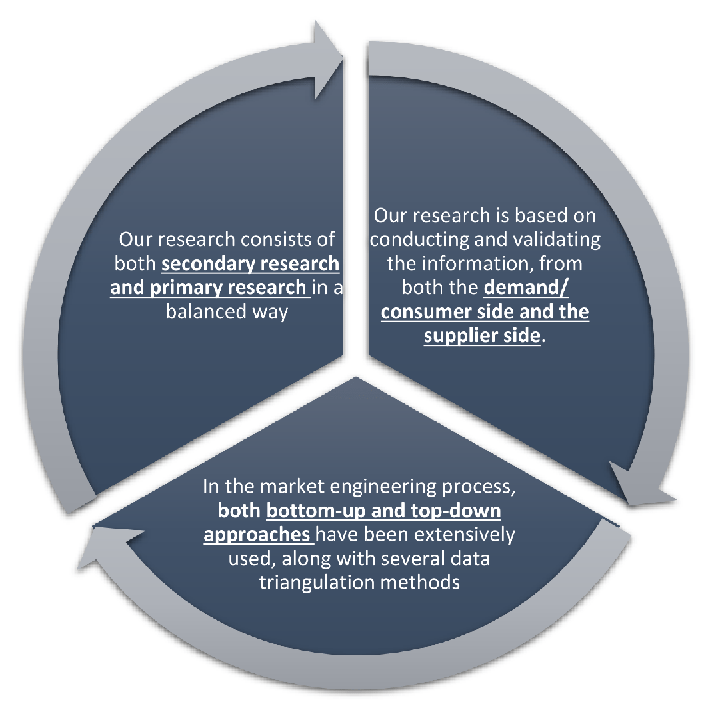
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
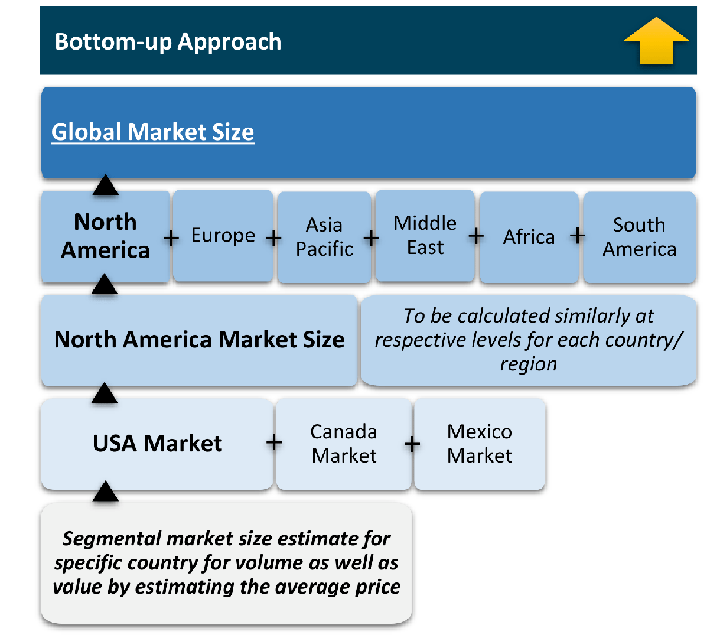
Research Approach
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
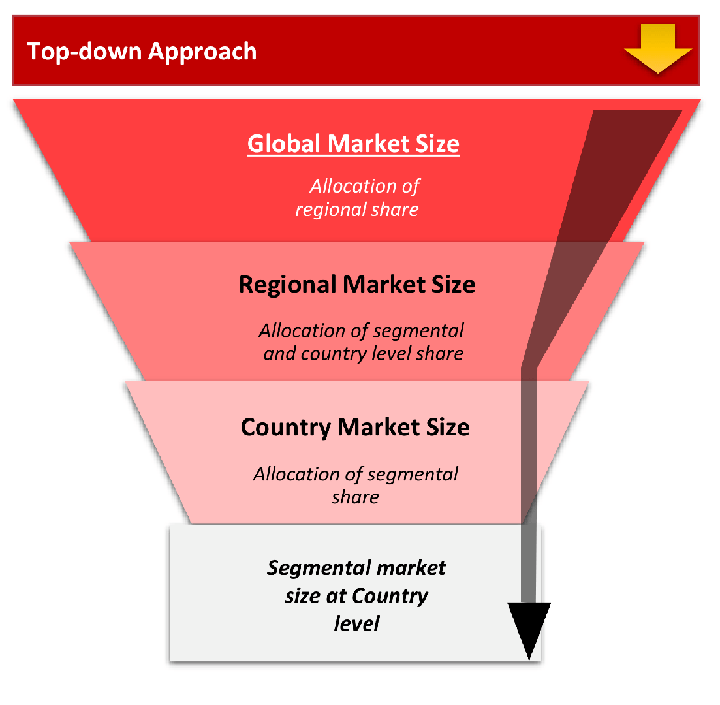
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
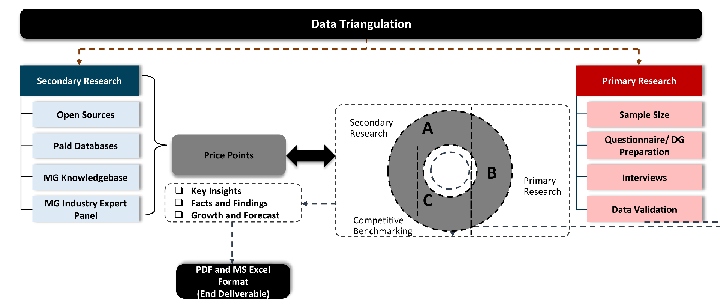
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation