Sleep Apnea Implants Market Size, Share & Trends Analysis Report by Device Type (Hypoglossal Nerve Stimulation (HNS) Devices, Soft Palate Implants, Tongue Base Suspension Devices, Combination Implant Systems, Others), Technology, Rated Power, Implant Material, Severity of Sleep Apnea, Patient Age Group, End-Use, and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Sleep Apnea Implants Market Size, Share, and Growth
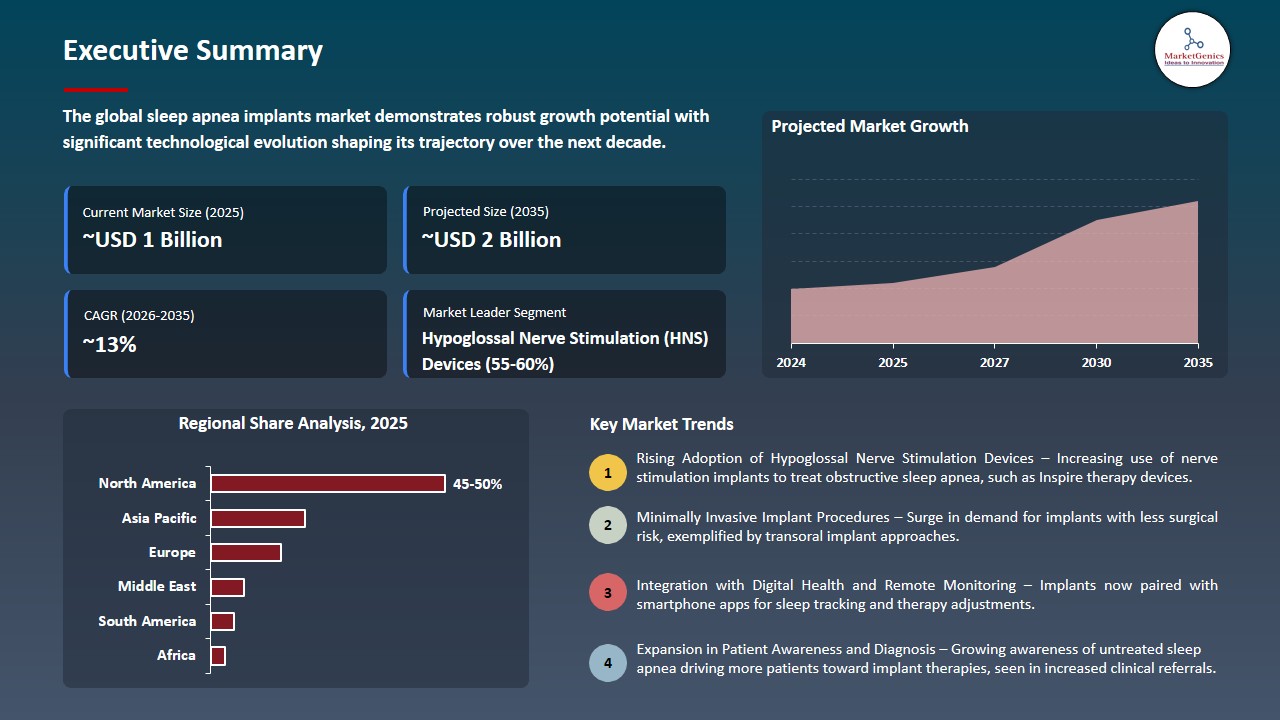
The global sleep apnea implants market is witnessing strong growth, valued at USD 0.6 billion in 2025 and projected to reach USD 1.9 billion by 2035, expanding at a CAGR of 12.5% during the forecast period. Asia Pacific is the fastest-growing region in the sleep apnea implants market due to rising awareness of sleep disorders, increasing diagnosis rates, a large untreated patient population, rapid urbanization and lifestyle changes, and improving access to advanced healthcare technologies supported by growing healthcare expenditure.
Olivier Taelman, Nyxoah’s Chief Executive Officer, said, “Today marks a defining moment for Nyxoah and for U.S. patients suffering from OSA. With the FDA’s marketing approval of the Genio system, we are proud to bring this innovative therapy to the U.S. market, our mission has always been to make sleep simple for OSA patients by offering them a solution that empowers better sleep. We look forward to the successful execution of our U.S. commercialization strategy”.
The growing prevalence of obstructive and central sleep apnea across the globe due to various reasons including increasing obesity rates, ageing and sedentary lifestyles are greatly broadening the pool of patients who will need the effective treatment. Since the increasing number of people have been diagnosed with moderate to severe sleep apnea, there is an increasing demand in the development of advanced and long-term management of sleep apnea like implantable devices, which put sleep apnea implants as a key solution in the management of this prevalent health.
The increased use of ambulatory and outpatient procedures also provides a great opportunity of the sleep apnea implants market. With the possibility to minimize the invasive nature of implantations in an outpatient facility, the health practitioners are likely to minimize the overall cost of treatment, shorten the overall patient recovery period, and help to increase the level of convenience and access. This pattern promotes the increased use of implant-based treatment, especially in those patients who want to have lasting and efficient solutions to the conventional in-hospital treatment.
Adjacent opportunities in the sleep apnea implants market involve the inclusion of digital health and remote monitoring technologies, creation of next generation neurostimulation systems with increased safety and comfort, expansion of minimally invasive outpatient procedures, optimization of personal therapies, and penetration into new markets with an increasing prevalence of OSA and an enhanced healthcare infrastructure, all of which enable further adoption and long-term expansion of implant-based sleep apnea therapies.

Sleep Apnea Implants Market Dynamics and Trends
Driver: Treatment-Resistant Cases & CPAP Intolerance
-
The high rate of cases that cannot be treated by the conventional methods and the common lack of intolerance to Continuous Positive Airway pressure (CPAP) therapy is another crucial market driver in the sleep apnea implants market. Most of the patients complain of discomfort, nasal congestion, irritation caused by the masks, or inability to stick to the daily use of CPAP leading to inadequate compliance, and the ineffective control of obstructive sleep apnea.
- The restrictions result in high demand of alternative therapies that offer effective and long-term relief of symptoms. Minimally invasive, consistently effective, and higher patient comfort, along with less reliant on bulky equipment, are all possible with implantable devices, like hypoglossal nerve stimulators.
- Inspire Medical Systems introduced its FDA-cleared Inspire V neurostimulation system in the U.S. in 2025, which provides a long-term moderate to severe obstructive sleep apnea minimally invasive alternative. Favorable feedbacks of surgeons and patients, good adoption, points out the leadership of the Inspire, and positive trends in the need to implantable sleep apnea therapies.
- The increased awareness of CPAP restrictions among patients and clinicians is fastening the uptake of sleep apnea implants around the world.
Restraint: Regulatory and Approval Challenges
-
The sleep apnea implants market is highly restrained by the tough regulatory and approval procedures in key markets. Any implantable devices, including hypoglossal nerve stimulators have to be subjected to extensive clinical trials to prove their safety and effectiveness, to conform to the high standards of quality, to meet the standards established by the regulatory agencies, including the U.S. FDA, European CE Mark, and other governmental organizations.
- These complicated approval processes may result in very long product development processes and late market introduction, restricting the rate at which new technologies are made available to patients. Moreover, it makes it harder to comply with the regulations, which augment the cost of development since manufacturers are required to spend a lot of money on clinical trials, documentation, and post-market observations programs.
- Together with the necessity of constant revisions to meet new standards, regulatory obstacles constitute one of the significant limitations on the development and expansion of the global sleep apnea implants market.
Opportunity: AI-Driven Personalization & Next-Gen Devices
-
The introduction of machine learning and artificial intelligence (AI) into sleep apnea implants is a great opportunity to grow the market. AI technologies can be used to administer automatic and customized forms of stimulation therapy that are based on the unique breathing patterns, sleep posture, and the level of obstructive sleep apnea in an individual patient. This individualization promotes therapeutic success, comfort, and compliance to long-term therapy, which is the greatest shortcoming of conventional therapies like CPAP.
- Next-generation implantable devices with AI can also be remote monitored, predictively analized, and automatically adjusted to their therapy to minimize the necessity to regularly visit the clinic and allow healthcare professionals to optimally adjust the treatment results.
- In December 2025, Capgemini introduced the first-of-its-kind AI-powered implantable sleep apnea, which customizes neurostimulation according to real-time sleep stages, breathing patterns, and body position. As opposed to CPAP, it can be implanted completely, making it much more comfortable and compliant, decreasing the rate of Apnea-Hypopnea Index (AHI) by a large margin and improving the overall sleep quality.
- These technological advances will likely spur uptake, diversify products in a competitive market and increase the scope of globally available implant-based sleep apnea treatment.
Key Trend: Digitally Integrated Implants
-
The implementation of digitally integrated sleep apnea devices will become an important trend due to the development of connectivity and smart health devices. Implants of modern times have Bluetooth connectivity, which allows smooth interaction with applications installed on the phone that display the usage of therapy, sleep patterns, and progress of a patient in real-time.
- At the same time, the physician dashboards in the cloud give clinicians access to patient data constantly, enabling remote access, adjustment of therapies, and optimization of treatment tailored to the patient. This combination improves patient interaction, compliance and facilitates proactive intervention which will minimize the complications that come with lack of treated sleep apnea.
- Inspire Medical Systems released the Inspire SleepSync system, a networked patient management system that incorporates a Bluetooth-enabled remote, a mobile application, and a web portal. The platform facilitates the wireless tracking of implants, simplified care management and increased patient interaction.
- Digitally integrated implants become a significant point of differentiation in healthcare as it goes more connected and data-driven, benefiting clinical outcomes and expanding the use of implant-based sleep apnea treatments.
Sleep-Apnea-Implants-Market Analysis and Segmental Data
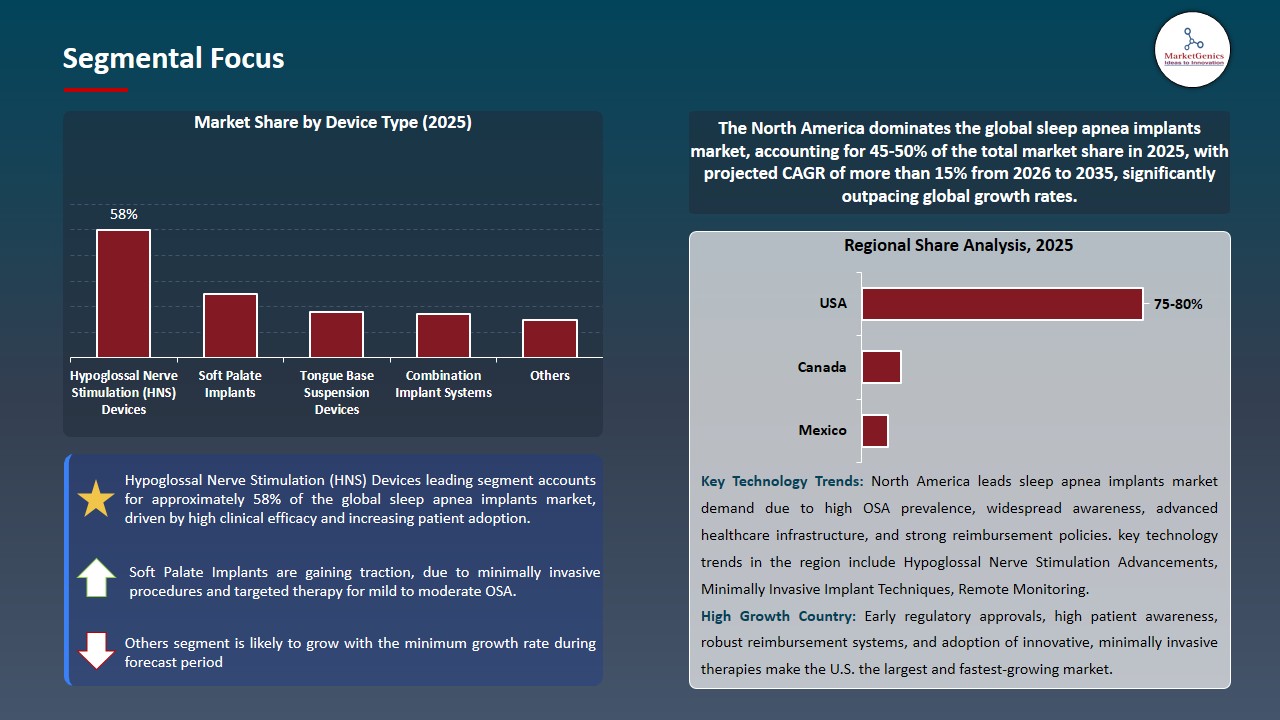
Hypoglossal Nerve Stimulation (HNS) Devices Dominate Global Sleep Apnea Implants Market
-
The hypoglossal nerve stimulation devices is the leading segment in the global sleep apnea implants market, holding the largest share due to its proven clinical effectiveness in treating moderate to severe obstructive sleep apnea. HNS devices work by stimulating the hypoglossal nerve to maintain airway patency during sleep, offering a minimally invasive and long-term alternative to conventional therapies such as CPAP, which often face low patient compliance.
- The segment’s dominance is further supported by increasing physician preference, growing patient acceptance, and continuous technological advancements that enhance safety, comfort, and device performance. Integration with remote monitoring and personalized therapy adjustment features also contributes to higher adoption rates, solidifying HNS devices as the preferred choice for sleep apnea implant therapy worldwide.
- On August 2025, Nyxoah received FDA approval for its Genio system, a hypoglossal nerve stimulator for patients with moderate to severe obstructive sleep apnea (AHI 15–65) who cannot tolerate or have failed CPAP therapy.
- Hypoglossal nerve stimulation devices remain the dominant and preferred segment in the global sleep apnea implants market due to their effectiveness, patient acceptance, and ongoing technological innovation.
North America Leads Global Sleep Apnea Implants Market Demand
-
North America is the leading region in the sleep apnea implants market, driven by a high prevalence of obstructive sleep apnea and growing awareness of its associated health risks, including cardiovascular disease, diabetes, and cognitive impairment.
- Patients and healthcare providers in the region are increasingly seeking effective, long-term alternatives to conventional therapies such as CPAP, which has a high rate of intolerance and non-compliance. This preference for implantable solutions is a major factor fueling market growth.
- The presence of advanced healthcare infrastructure, well-trained ENT surgeons and sleep specialists, and widespread access to diagnostic and treatment facilities further supports the adoption of implant-based therapies. In addition, favorable reimbursement policies and insurance coverage for surgical interventions lower financial barriers, encouraging more patients to opt for these solutions.
- Ongoing technological advancements, such as hypoglossal nerve stimulation devices with improved safety, comfort, and remote monitoring capabilities, are also contributing to North America’s leadership in the global sleep apnea implants market, positioning the region as a key driver of overall market growth.
- North America’s combination of high disease prevalence, advanced healthcare infrastructure, patient awareness, and technological innovation firmly establishes it as the leading driver of growth in the global sleep apnea implants market.
Sleep-Apnea-Implants-Market Ecosystem
The global sleep apnea implants market is moderately consolidated, with key players including Inspire Medical Systems, Respicardia, Inc., Medtronic Plc, Nyxoah S.A., and LivaNova PLC. These companies maintain competitive strength through strong investments in research and development, advanced neurostimulation technologies, clinically proven implant systems, and well-established relationships with sleep specialists, ENT surgeons, and healthcare institutions. Their market presence is further supported by focused product portfolios, physician training initiatives, and growing adoption of implant-based alternatives to CPAP therapy.
The sleep apnea implants value chain encompasses raw material sourcing and component manufacturing, device design and assembly under stringent quality and safety standards, clinical trials and regulatory approvals, product branding and packaging, and distribution through hospitals, sleep centers, and specialized clinics. Post-market activities include physician education, patient monitoring, therapy optimization, and long-term device management.
Entry barriers remain high due to rigorous regulatory requirements, the need for extensive clinical validation, specialized surgical expertise, technological complexity, and strong competition from established manufacturers. Continuous innovation in minimally invasive implantation techniques, device miniaturization, personalized stimulation therapies, and integration with digital monitoring platforms continues to drive differentiation and adoption across the global market.

Recent Development and Strategic Overview:
-
In May 2025, LivaNova completed the FDA premarket approval submission for its aura6000 System, a hypoglossal nerve stimulation implant for obstructive sleep apnea. The submission was supported by 12-month OSPREY trial data showing a 65% responder rate, 68% median reduction in AHI, and durable therapeutic impact, highlighting the system’s effectiveness for patients with severe OSA and high-risk airway collapse.
- In December 2025, Resmed received FDA clearance for its AI-enabled Smart Comfort system, designed to personalize CPAP therapy for patients with obstructive sleep apnea. Leveraging machine learning and real-world sleep data, the device recommends individualized comfort settings, improving adherence, patient engagement, and therapy outcomes, and exemplifying the integration of AI and digital health in sleep care.
Report Scope
|
Detail |
|
|
Market Size in 2025 |
USD 0.6 Bn |
|
Market Forecast Value in 2035 |
USD 1.9 Bn |
|
Growth Rate (CAGR) |
12.5% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
US$ Billion for Value Thousand Units for Volume |
|
Report Format |
Electronic (PDF) + Excel |
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Sleep-Apnea-Implants-Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Sleep Apnea Implants Market, By Device Type |
|
|
Sleep Apnea Implants Market, By Technology |
|
|
Sleep Apnea Implants Market, By Rated Power |
|
|
Sleep Apnea Implants Market, By Implant Material |
|
|
Sleep Apnea Implants Market, By Severity of Sleep Apnea |
|
|
Sleep Apnea Implants Market, By Patient Age Group |
|
|
Sleep Apnea Implants Market, By End-Use |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Sleep Apnea Implants Market Outlook
- 2.1.1. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Sleep Apnea Implants Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Industry Ecosystem Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.4.1. Import & Export Analysis, 2025
- 3.4.2. Top Importing Countries
- 3.4.3. Top Exporting Countries
- 3.5. Trump Tariff Impact Analysis
- 3.5.1. Manufacturer
- 3.5.1.1. Based on the component & Raw material
- 3.5.2. Supply Chain
- 3.5.3. End Consumer
- 3.5.1. Manufacturer
- 3.6. Raw Material Analysis
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising prevalence and diagnosis of sleep apnea
- 4.1.1.2. Advances in implantable neurostimulation and minimally invasive devices
- 4.1.1.3. Preference for long-term alternatives to CPAP therapy
- 4.1.2. Restraints
- 4.1.2.1. High cost of implants and surgical procedures
- 4.1.2.2. Limited awareness, reimbursement challenges, and regulatory barriers.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.4.1. Component Suppliers
- 4.4.2. Device Manufacturers
- 4.4.3. Distribution & Sales Channels
- 4.4.4. Healthcare Providers
- 4.4.5. End Users/Patients
- 4.5. Porter’s Five Forces Analysis
- 4.6. PESTEL Analysis
- 4.7. Global Sleep Apnea Implants Market Demand
- 4.7.1. Historical Market Size – Volume (Thousand Units) and Value (US$ Bn), 2020-2024
- 4.7.2. Current and Future Market Size – Volume (Thousand Units) and Value (US$ Bn), 2026–2035
- 4.7.2.1. Y-o-Y Growth Trends
- 4.7.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Sleep Apnea Implants Market Analysis, by Device Type
- 6.1. Key Segment Analysis
- 6.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Product Type, 2021-2035
- 6.2.1. Hypoglossal Nerve Stimulation (HNS) Devices
- 6.2.2. Soft Palate Implants
- 6.2.3. Tongue Base Suspension Devices
- 6.2.4. Combination Implant Systems
- 6.2.5. Others
- 7. Global Sleep Apnea Implants Market Analysis, by Technology
- 7.1. Key Segment Analysis
- 7.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Technology, 2021-2035
- 7.2.1. Neurostimulation Technology
- 7.2.2. Implantable Pulse Generator (IPG) Systems
- 7.2.3. Sensing-Enabled Devices
- 7.2.4. Battery-Powered Implants
- 7.2.5. Rechargeable Implant Systems
- 7.2.6. Others
- 8. Global Sleep Apnea Implants Market Analysis, by Rated Power
- 8.1. Key Segment Analysis
- 8.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Rated Power, 2021-2035
- 8.2.1. Less than 0.5W
- 8.2.2. 5W - 1.5W
- 8.2.3. Above 1.5W
- 9. Global Sleep Apnea Implants Market Analysis, by Implant Material
- 9.1. Key Segment Analysis
- 9.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Implant Material, 2021-2035
- 9.2.1. Titanium-Based Implants
- 9.2.2. Biocompatible Polymer Implants
- 9.2.3. Silicone-Based Implants
- 9.2.4. Hybrid Material Implants
- 10. Global Sleep Apnea Implants Market Analysis, by Severity of Sleep Apnea
- 10.1. Key Segment Analysis
- 10.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Severity of Sleep Apnea, 2021-2035
- 10.2.1. AHI 5-15
- 10.2.2. AHI 15-30
- 10.2.3. AHI > 30
- 11. Global Sleep Apnea Implants Market Analysis, by Patient Age Group
- 11.1. Key Segment Analysis
- 11.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Patient Age Group, 2021-2035
- 11.2.1. Pediatric (< 18 Years)
- 11.2.2. Adult (18-65 Years)
- 11.2.3. Geriatric (> 65 Years)
- 12. Global Sleep Apnea Implants Market Analysis and Forecasts, by End-use
- 12.1. Key Findings
- 12.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by End-use, 2021-2035
- 12.2.1. Hospitals
- 12.2.2. Specialty Clinics
- 12.2.3. Ambulatory Surgical Centers
- 12.2.4. Home Healthcare
- 12.2.5. Rehabilitation Centers
- 12.2.6. Others
- 13. Global Sleep Apnea Implants Market Analysis and Forecasts, by Region
- 13.1. Key Findings
- 13.2. Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 13.2.1. North America
- 13.2.2. Europe
- 13.2.3. Asia Pacific
- 13.2.4. Middle East
- 13.2.5. Africa
- 13.2.6. South America
- 14. North America Sleep Apnea Implants Market Analysis
- 14.1. Key Segment Analysis
- 14.2. Regional Snapshot
- 14.3. North America Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 14.3.1. Device Type
- 14.3.2. Technology
- 14.3.3. Rated Power
- 14.3.4. Implant Material
- 14.3.5. Severity of Sleep Apnea
- 14.3.6. Patient Age Group
- 14.3.7. End-Use
- 14.3.8. Country
- 14.3.8.1. USA
- 14.3.8.2. Canada
- 14.3.8.3. Mexico
- 14.4. USA Sleep Apnea Implants Market
- 14.4.1. Country Segmental Analysis
- 14.4.2. Device Type
- 14.4.3. Technology
- 14.4.4. Rated Power
- 14.4.5. Implant Material
- 14.4.6. Severity of Sleep Apnea
- 14.4.7. Patient Age Group
- 14.4.8. End-Use
- 14.5. Canada Sleep Apnea Implants Market
- 14.5.1. Country Segmental Analysis
- 14.5.2. Device Type
- 14.5.3. Technology
- 14.5.4. Rated Power
- 14.5.5. Implant Material
- 14.5.6. Severity of Sleep Apnea
- 14.5.7. Patient Age Group
- 14.5.8. End-Use
- 14.6. Mexico Sleep Apnea Implants Market
- 14.6.1. Country Segmental Analysis
- 14.6.2. Device Type
- 14.6.3. Technology
- 14.6.4. Rated Power
- 14.6.5. Implant Material
- 14.6.6. Severity of Sleep Apnea
- 14.6.7. Patient Age Group
- 14.6.8. End-Use
- 15. Europe Sleep Apnea Implants Market Analysis
- 15.1. Key Segment Analysis
- 15.2. Regional Snapshot
- 15.3. Europe Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 15.3.1. Device Type
- 15.3.2. Technology
- 15.3.3. Rated Power
- 15.3.4. Implant Material
- 15.3.5. Severity of Sleep Apnea
- 15.3.6. Patient Age Group
- 15.3.7. End-Use
- 15.3.8. Country
- 15.3.8.1. Germany
- 15.3.8.2. United Kingdom
- 15.3.8.3. France
- 15.3.8.4. Italy
- 15.3.8.5. Spain
- 15.3.8.6. Netherlands
- 15.3.8.7. Nordic Countries
- 15.3.8.8. Poland
- 15.3.8.9. Russia & CIS
- 15.3.8.10. Rest of Europe
- 15.4. Germany Sleep Apnea Implants Market
- 15.4.1. Country Segmental Analysis
- 15.4.2. Device Type
- 15.4.3. Technology
- 15.4.4. Rated Power
- 15.4.5. Implant Material
- 15.4.6. Severity of Sleep Apnea
- 15.4.7. Patient Age Group
- 15.4.8. End-Use
- 15.5. United Kingdom Sleep Apnea Implants Market
- 15.5.1. Country Segmental Analysis
- 15.5.2. Device Type
- 15.5.3. Technology
- 15.5.4. Rated Power
- 15.5.5. Implant Material
- 15.5.6. Severity of Sleep Apnea
- 15.5.7. Patient Age Group
- 15.5.8. End-Use
- 15.6. France Sleep Apnea Implants Market
- 15.6.1. Country Segmental Analysis
- 15.6.2. Device Type
- 15.6.3. Technology
- 15.6.4. Rated Power
- 15.6.5. Implant Material
- 15.6.6. Severity of Sleep Apnea
- 15.6.7. Patient Age Group
- 15.6.8. End-Use
- 15.7. Italy Sleep Apnea Implants Market
- 15.7.1. Country Segmental Analysis
- 15.7.2. Device Type
- 15.7.3. Technology
- 15.7.4. Rated Power
- 15.7.5. Implant Material
- 15.7.6. Severity of Sleep Apnea
- 15.7.7. Patient Age Group
- 15.7.8. End-Use
- 15.8. Spain Sleep Apnea Implants Market
- 15.8.1. Country Segmental Analysis
- 15.8.2. Device Type
- 15.8.3. Technology
- 15.8.4. Rated Power
- 15.8.5. Implant Material
- 15.8.6. Severity of Sleep Apnea
- 15.8.7. Patient Age Group
- 15.8.8. End-Use
- 15.9. Netherlands Sleep Apnea Implants Market
- 15.9.1. Country Segmental Analysis
- 15.9.2. Device Type
- 15.9.3. Technology
- 15.9.4. Rated Power
- 15.9.5. Implant Material
- 15.9.6. Severity of Sleep Apnea
- 15.9.7. Patient Age Group
- 15.9.8. End-Use
- 15.10. Nordic Countries Sleep Apnea Implants Market
- 15.10.1. Country Segmental Analysis
- 15.10.2. Device Type
- 15.10.3. Technology
- 15.10.4. Rated Power
- 15.10.5. Implant Material
- 15.10.6. Severity of Sleep Apnea
- 15.10.7. Patient Age Group
- 15.10.8. End-Use
- 15.11. Poland Sleep Apnea Implants Market
- 15.11.1. Country Segmental Analysis
- 15.11.2. Device Type
- 15.11.3. Technology
- 15.11.4. Rated Power
- 15.11.5. Implant Material
- 15.11.6. Severity of Sleep Apnea
- 15.11.7. Patient Age Group
- 15.11.8. End-Use
- 15.12. Russia & CIS Sleep Apnea Implants Market
- 15.12.1. Country Segmental Analysis
- 15.12.2. Device Type
- 15.12.3. Technology
- 15.12.4. Rated Power
- 15.12.5. Implant Material
- 15.12.6. Severity of Sleep Apnea
- 15.12.7. Patient Age Group
- 15.12.8. End-Use
- 15.13. Rest of Europe Sleep Apnea Implants Market
- 15.13.1. Country Segmental Analysis
- 15.13.2. Device Type
- 15.13.3. Technology
- 15.13.4. Rated Power
- 15.13.5. Implant Material
- 15.13.6. Severity of Sleep Apnea
- 15.13.7. Patient Age Group
- 15.13.8. End-Use
- 16. Asia Pacific Sleep Apnea Implants Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. Asia Pacific Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Device Type
- 16.3.2. Technology
- 16.3.3. Rated Power
- 16.3.4. Implant Material
- 16.3.5. Severity of Sleep Apnea
- 16.3.6. Patient Age Group
- 16.3.7. End-Use
- 16.3.8. Country
- 16.3.8.1. China
- 16.3.8.2. India
- 16.3.8.3. Japan
- 16.3.8.4. South Korea
- 16.3.8.5. Australia and New Zealand
- 16.3.8.6. Indonesia
- 16.3.8.7. Malaysia
- 16.3.8.8. Thailand
- 16.3.8.9. Vietnam
- 16.3.8.10. Rest of Asia Pacific
- 16.4. China Sleep Apnea Implants Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Device Type
- 16.4.3. Technology
- 16.4.4. Rated Power
- 16.4.5. Implant Material
- 16.4.6. Severity of Sleep Apnea
- 16.4.7. Patient Age Group
- 16.4.8. End-Use
- 16.5. India Sleep Apnea Implants Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Device Type
- 16.5.3. Technology
- 16.5.4. Rated Power
- 16.5.5. Implant Material
- 16.5.6. Severity of Sleep Apnea
- 16.5.7. Patient Age Group
- 16.5.8. End-Use
- 16.6. Japan Sleep Apnea Implants Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Device Type
- 16.6.3. Technology
- 16.6.4. Rated Power
- 16.6.5. Implant Material
- 16.6.6. Severity of Sleep Apnea
- 16.6.7. Patient Age Group
- 16.6.8. End-Use
- 16.7. South Korea Sleep Apnea Implants Market
- 16.7.1. Country Segmental Analysis
- 16.7.2. Device Type
- 16.7.3. Technology
- 16.7.4. Rated Power
- 16.7.5. Implant Material
- 16.7.6. Severity of Sleep Apnea
- 16.7.7. Patient Age Group
- 16.7.8. End-Use
- 16.8. Australia and New Zealand Sleep Apnea Implants Market
- 16.8.1. Country Segmental Analysis
- 16.8.2. Device Type
- 16.8.3. Technology
- 16.8.4. Rated Power
- 16.8.5. Implant Material
- 16.8.6. Severity of Sleep Apnea
- 16.8.7. Patient Age Group
- 16.8.8. End-Use
- 16.9. Indonesia Sleep Apnea Implants Market
- 16.9.1. Country Segmental Analysis
- 16.9.2. Device Type
- 16.9.3. Technology
- 16.9.4. Rated Power
- 16.9.5. Implant Material
- 16.9.6. Severity of Sleep Apnea
- 16.9.7. Patient Age Group
- 16.9.8. End-Use
- 16.10. Malaysia Sleep Apnea Implants Market
- 16.10.1. Country Segmental Analysis
- 16.10.2. Device Type
- 16.10.3. Technology
- 16.10.4. Rated Power
- 16.10.5. Implant Material
- 16.10.6. Severity of Sleep Apnea
- 16.10.7. Patient Age Group
- 16.10.8. End-Use
- 16.11. Thailand Sleep Apnea Implants Market
- 16.11.1. Country Segmental Analysis
- 16.11.2. Device Type
- 16.11.3. Technology
- 16.11.4. Rated Power
- 16.11.5. Implant Material
- 16.11.6. Severity of Sleep Apnea
- 16.11.7. Patient Age Group
- 16.11.8. End-Use
- 16.12. Vietnam Sleep Apnea Implants Market
- 16.12.1. Country Segmental Analysis
- 16.12.2. Device Type
- 16.12.3. Technology
- 16.12.4. Rated Power
- 16.12.5. Implant Material
- 16.12.6. Severity of Sleep Apnea
- 16.12.7. Patient Age Group
- 16.12.8. End-Use
- 16.13. Rest of Asia Pacific Sleep Apnea Implants Market
- 16.13.1. Country Segmental Analysis
- 16.13.2. Device Type
- 16.13.3. Technology
- 16.13.4. Rated Power
- 16.13.5. Implant Material
- 16.13.6. Severity of Sleep Apnea
- 16.13.7. Patient Age Group
- 16.13.8. End-Use
- 17. Middle East Sleep Apnea Implants Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Middle East Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Device Type
- 17.3.2. Technology
- 17.3.3. Rated Power
- 17.3.4. Implant Material
- 17.3.5. Severity of Sleep Apnea
- 17.3.6. Patient Age Group
- 17.3.7. End-Use
- 17.3.8. Country
- 17.3.8.1. Turkey
- 17.3.8.2. UAE
- 17.3.8.3. Saudi Arabia
- 17.3.8.4. Israel
- 17.3.8.5. Rest of Middle East
- 17.4. Turkey Sleep Apnea Implants Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Device Type
- 17.4.3. Technology
- 17.4.4. Rated Power
- 17.4.5. Implant Material
- 17.4.6. Severity of Sleep Apnea
- 17.4.7. Patient Age Group
- 17.4.8. End-Use
- 17.5. UAE Sleep Apnea Implants Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Device Type
- 17.5.3. Technology
- 17.5.4. Rated Power
- 17.5.5. Implant Material
- 17.5.6. Severity of Sleep Apnea
- 17.5.7. Patient Age Group
- 17.5.8. End-Use
- 17.6. Saudi Arabia Sleep Apnea Implants Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Device Type
- 17.6.3. Technology
- 17.6.4. Rated Power
- 17.6.5. Implant Material
- 17.6.6. Severity of Sleep Apnea
- 17.6.7. Patient Age Group
- 17.6.8. End-Use
- 17.7. Israel Sleep Apnea Implants Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Device Type
- 17.7.3. Technology
- 17.7.4. Rated Power
- 17.7.5. Implant Material
- 17.7.6. Severity of Sleep Apnea
- 17.7.7. Patient Age Group
- 17.7.8. End-Use
- 17.8. Rest of Middle East Sleep Apnea Implants Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Device Type
- 17.8.3. Technology
- 17.8.4. Rated Power
- 17.8.5. Implant Material
- 17.8.6. Severity of Sleep Apnea
- 17.8.7. Patient Age Group
- 17.8.8. End-Use
- 18. Africa Sleep Apnea Implants Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Africa Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Device Type
- 18.3.2. Technology
- 18.3.3. Rated Power
- 18.3.4. Implant Material
- 18.3.5. Severity of Sleep Apnea
- 18.3.6. Patient Age Group
- 18.3.7. End-Use
- 18.3.8. Country
- 18.3.8.1. South Africa
- 18.3.8.2. Egypt
- 18.3.8.3. Nigeria
- 18.3.8.4. Algeria
- 18.3.8.5. Rest of Africa
- 18.4. South Africa Sleep Apnea Implants Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Device Type
- 18.4.3. Technology
- 18.4.4. Rated Power
- 18.4.5. Implant Material
- 18.4.6. Severity of Sleep Apnea
- 18.4.7. Patient Age Group
- 18.4.8. End-Use
- 18.5. Egypt Sleep Apnea Implants Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Device Type
- 18.5.3. Technology
- 18.5.4. Rated Power
- 18.5.5. Implant Material
- 18.5.6. Severity of Sleep Apnea
- 18.5.7. Patient Age Group
- 18.5.8. End-Use
- 18.6. Nigeria Sleep Apnea Implants Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Device Type
- 18.6.3. Technology
- 18.6.4. Rated Power
- 18.6.5. Implant Material
- 18.6.6. Severity of Sleep Apnea
- 18.6.7. Patient Age Group
- 18.6.8. End-Use
- 18.7. Algeria Sleep Apnea Implants Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Device Type
- 18.7.3. Technology
- 18.7.4. Rated Power
- 18.7.5. Implant Material
- 18.7.6. Severity of Sleep Apnea
- 18.7.7. Patient Age Group
- 18.7.8. End-Use
- 18.8. Rest of Africa Sleep Apnea Implants Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Device Type
- 18.8.3. Technology
- 18.8.4. Rated Power
- 18.8.5. Implant Material
- 18.8.6. Severity of Sleep Apnea
- 18.8.7. Patient Age Group
- 18.8.8. End-Use
- 19. South America Sleep Apnea Implants Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. South America Sleep Apnea Implants Market Size Volume (Thousand Units) and Value (US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Device Type
- 19.3.2. Technology
- 19.3.3. Rated Power
- 19.3.4. Implant Material
- 19.3.5. Severity of Sleep Apnea
- 19.3.6. Patient Age Group
- 19.3.7. End-Use
- 19.3.8. Country
- 19.3.8.1. Brazil
- 19.3.8.2. Argentina
- 19.3.8.3. Rest of South America
- 19.4. Brazil Sleep Apnea Implants Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Device Type
- 19.4.3. Technology
- 19.4.4. Rated Power
- 19.4.5. Implant Material
- 19.4.6. Severity of Sleep Apnea
- 19.4.7. Patient Age Group
- 19.4.8. End-Use
- 19.5. Argentina Sleep Apnea Implants Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Device Type
- 19.5.3. Technology
- 19.5.4. Rated Power
- 19.5.5. Implant Material
- 19.5.6. Severity of Sleep Apnea
- 19.5.7. Patient Age Group
- 19.5.8. End-Use
- 19.6. Rest of South America Sleep Apnea Implants Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Device Type
- 19.6.3. Technology
- 19.6.4. Rated Power
- 19.6.5. Implant Material
- 19.6.6. Severity of Sleep Apnea
- 19.6.7. Patient Age Group
- 19.6.8. End-Use
- 20. Key Players/ Company Profile
- 20.1. Abbott Laboratories
- 20.1.1. Company Details/ Overview
- 20.1.2. Company Financials
- 20.1.3. Key Customers and Competitors
- 20.1.4. Business/ Industry Portfolio
- 20.1.5. Product Portfolio/ Specification Details
- 20.1.6. Pricing Data
- 20.1.7. Strategic Overview
- 20.1.8. Recent Developments
- 20.2. Advance Bio Medical Devices
- 20.3. Apnex Medical
- 20.4. Boston Scientific Corporation
- 20.5. Fisher & Paykel Healthcare
- 20.6. ImThera Medical
- 20.7. Inspire Medical Systems
- 20.8. Johnson & Johnson (Ethicon)
- 20.9. Linguaflex
- 20.10. LivaNova
- 20.11. Medihale Corporation
- 20.12. Medtronic
- 20.13. Neurent Medical
- 20.14. Nyxoah
- 20.15. Philips Healthcare
- 20.16. ResMed
- 20.17. ReVENT Medical
- 20.18. Siesta Medical
- 20.19. SomnoMed
- 20.20. Upper Airway Stimulation Therapeutics
- 20.21. Other Key Players
- 20.1. Abbott Laboratories
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
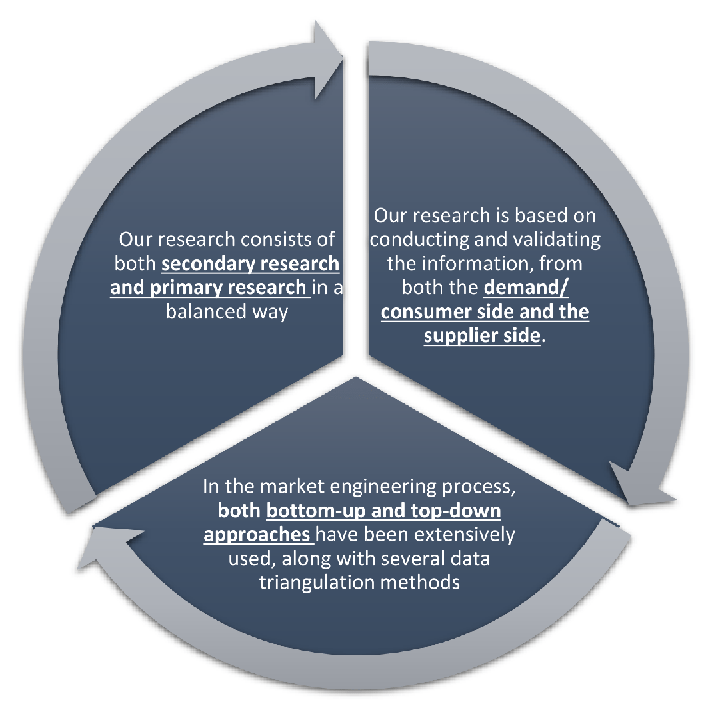
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
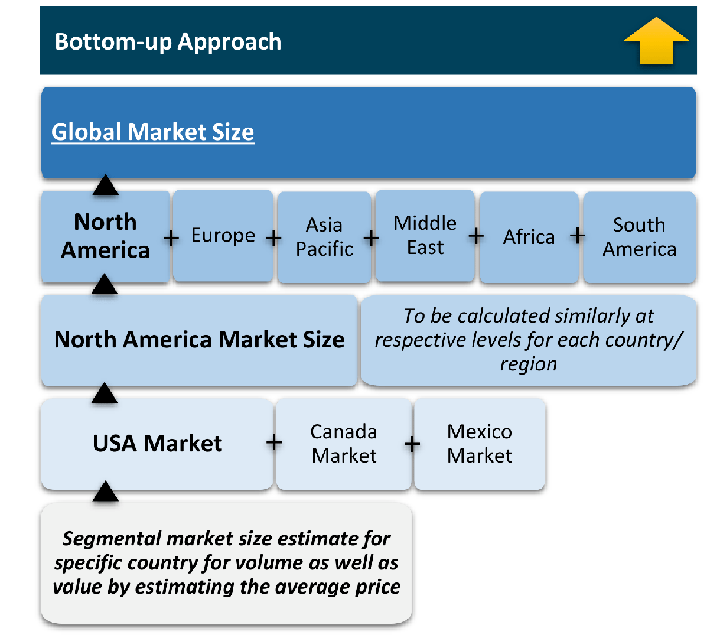
Research Approach
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation