Stem Cell Therapy Market Size, Share & Trends Analysis Report by Cell Source (Autologous Stem Cells, Allogeneic Stem Cells, Syngeneic Stem Cells), Cell Type, Technology, Therapeutic Application, Treatment Approach, End-Use, and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Stem Cell Therapy Market Size, Share, and Growth
The global stem cell therapy market is experiencing robust growth, with its estimated value of USD 0.6 billion in the year 2025 and ~USD 3 billion by 2035, registering a CAGR of 17.4%, during the forecast period. The global stem cell therapy market is majorly driven by rising chronic diseases, increasing geriatric population, advancements in regenerative medicine, supportive government initiatives, and growing awareness of stem cell treatments’ potential to repair or replace damaged tissues.

Dr. Tong Young Lee, CEO of StemCyte, said, "With one of the largest inventories of cord blood stem cells worldwide, StemCyte is uniquely positioned to supply, manufacture, and develop advanced cell therapy products, Looking ahead, we are committed to driving the development of innovative therapies, achieving sustainable growth through diversified business strategies, and solidifying our position as a global leader in the field of cell therapy."
The rising cost of chronic illnesses (cardiovascular, neurological, musculoskeletal disorders) and the aging population, which are becoming more and more probably seeking curative and minimally invasive treatment and care delivery methods, are shifting the need to stem cell therapies as an option to traditional methods of care delivery and treatment. For instance, in 2025, BrainStorm Cell Therapeutics receiving FDA approval to begin a Phase 3b trial of its adult stem cell therapy NurOwn to treat ALS, showing clinical momentum in using stem cell-based strategies to treat hard-to-treat neurodegenerative diseases. The growing clinical usage is driving market growth through matching breakthrough therapies with unmet health care requirements and patient outcomes.
Additionally, the large-scale investments, and corporate strategic programs are supporting the innovation and commercialization of stem cell technology. For instance, in November 2025, Klotho Neurosciences will receive the BioTech Breakthrough Cell Therapy Innovation of the Year award, which marks progress in the platform and its opportunity to expand indications and invest. The ability to invest and innovate intensifies pipeline growth, which enhances business opportunities and market growth opportunities.
Adjacent opportunities for the stem cell therapy market include the regenerative medicine market, tissue engineering and 3D bioprinting, gene-editing and CRISPR-based therapies, biobanking and cell preservation services, and advanced immunotherapies such as CAR-T cell therapy all leveraging stem cell science to expand therapeutic frontiers. These adjacent markets enhance revenue bases, speed up innovation and enhance growth and clinical applicability of the stem cell therapy ecosystem.

Stem Cell Therapy Market Dynamics and Trends
Driver: Strategic Commercial Launch of Medical-Grade Stem Cell Anti-Aging Product Lines by Key Innovators
-
The emerging market in the stem cell therapeutics sector is also propelled by commercialization of new, consumer-focused stem cell products that go beyond the traditional clinical indications to higher-end regenerative and cosmetic medical uses. The change is connected with the increase in the demand towards preventive, longevity, and wellness-based therapeutic solutions.
- The efforts of the major stakeholders indicate the increasing conflux of regenerative medicine with aesthetic and lifestyle medical services. For instance, in November 2025 Visionary Holdings Inc. via its subsidiary Visionary Health Technology Group announced the availability of a medical grade stem cell anti-aging line of products targeting mainland China and other Asian markets, with custom mesenchymal stem cell-based and NK cell-based regenerative solutions.
- This action indicates a change in hospital-based therapies to more extensive longevity-based systems, reaching more patients, adopting providers, and commercialization avenues.
- This strategic product commercialization expands the market, creates demand in related regenerative medicine markets and speeds up revenue diversification among industry members.
Restraint: Regulatory and Clinical Evidence Challenges for Novel Stem Cell Therapies
-
The high level of scientific development, even though quite strict regulatory conditions and the necessity to provide strong clinical evidence can still be considered as the major limiting factors of the market expansion, especially of the innovative stem cell treatment that can find the popularization. Regulators in all regions have been using a cautious review standard to maintain patient safety, long-term effectiveness and consistency in manufacturing.
- Numerous breakthrough stem cell products are still under late-stage clinical testing and may not get conditional approvals or expanded indications without the generation of sufficient data. Despite the development of induced pluripotent stem cell (iPSC) and allogeneic strategies, development timelines can become long and slow time-to-market, which can be a burden to development budgets.
- Regulatory uncertainties and variations in international systems of approval are an additional constraint on the commercial scale-up of commercial activities and can deter an investment in high-risk therapeutic innovation particularly by smaller developers.
- This regulatory restraint slows adoption of therapeutics and helps to mitigate short term market growth by increasing their commercialization stages.
Opportunity: Nationwide Stem Cell Extraction and Cryogenic Storage Platform Expansion
-
The development of integrated national platforms that integrate stem cell extraction, cryogenic storage, processing, and clinical delivery is an emerging opportunity in the stem cell therapy ecosystem. These platforms make the treatment more accessible to patients, can lead to a higher long-term expectation of stem cell treatment, and can serve as the answer to the increasing number of people seeking regenerative medicine and longevity-centered interventions.
- In December 2025, EUDA Health Holdings opened a national stem cell harvesting, cryogenic storage and clinical delivery system in China in collaboration with Shenzhen Inno Immune and Wuhan Kaien Hospital. These efforts allow scalable, quality-controlled, and cell preservation and regenerative treatment accessibility over a wide geographic connection, which improves the effectiveness of supply chains and clinical preparedness as personalized and preventive medicine continues to gain traction.
- Growth of nationwide clinical and storage networks enhances a better market penetration and a scaled base on future innovation of therapeutic approach and personalized care.
Key Trend: Increasing Industry Collaborations and Licensing Agreements for Stem Cell Technology Platforms
-
The expansion of strategic partnerships and licensing and technology deals will impact the future of stem cell therapy as a major trend that will hasten commercialization and increase therapeutic pipeline diversities. These partnerships allow firms to pool together complementary scientific know-how and commercial strengths, shortening development cycles and enhancing market access to novel stem cell therapies.
- In October 2025, Visionary Holdings achieves a worldwide product and technology licensing deal with Jiangsu Yike Regenerative Medicine, giving it the rights globally to utilize the stem cell technology platform, which Yike operates, to cure diabetes remission as well as other indications of regeneration. This venture is indicative of a wider industry trend in the direction of mutual commercialization risk and portfolio diversification without necessarily having to shoulder the entire weight of in-house research and development.
- The collaborative approach to licensing enhances the speed of technology transfer, optimizes resource utilization, and broadens the range of stem cell therapeutic innovations available to patients worldwide.
Stem-Cell-Therapy-Market Analysis and Segmental Data
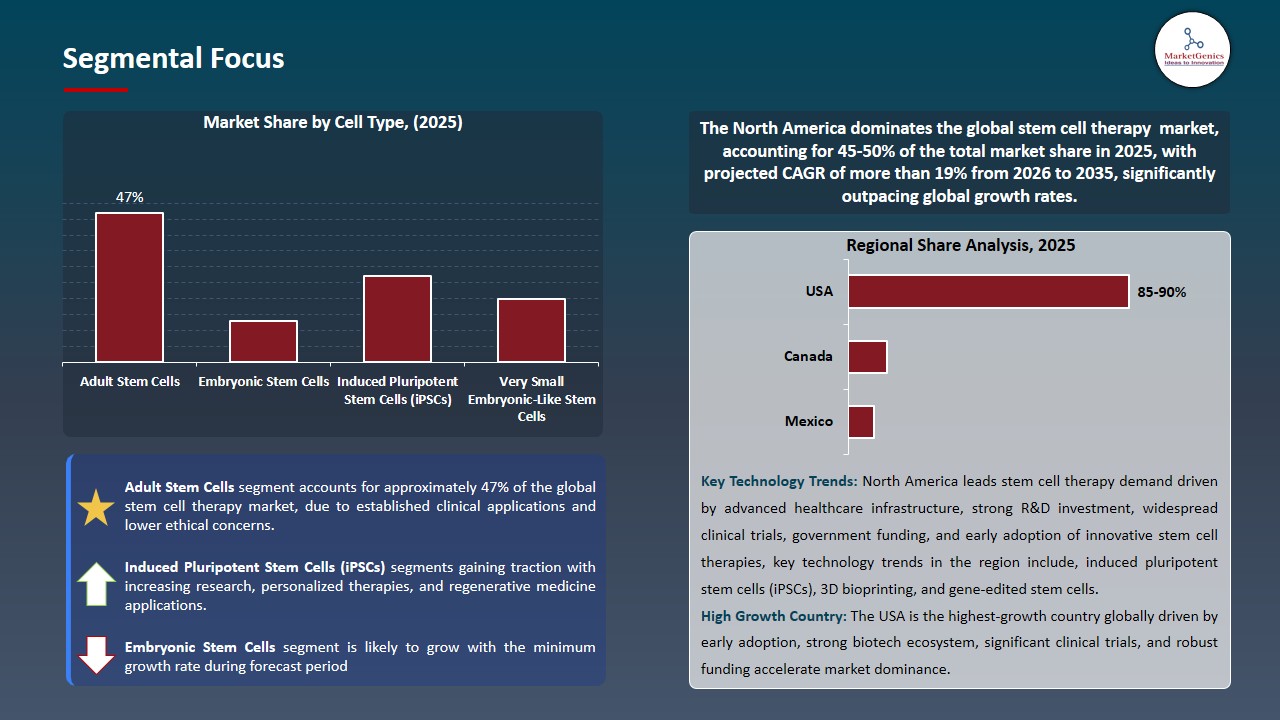
Adult Stem Cells Dominate Global Stem Cell Therapy Market
-
The adult stem cells segment dominates the global stem cell therapy market, the demonstrated safety track record, reduced ethical consideration, and effectiveness in the treatment of various disorders such as blood disorders, orthopedic injuries and autoimmune disorders among others. Hematopoietic stem cells (HSCs) and mesenchymal stem cells (MSCs) are the most widely used adult stem cells, supported by extensive clinical data and regulatory approvals.
- For instance, The FDA approved Mesoblast Ltd.'s Remestemcel-L, an MSC-based treatment, in 2024 to treat children steroid-refractory acute graft-versus-host disease, demonstrating the therapeutic and commercial potential of adult stem cells. Their availability, reduced chances of immune rejection and well-developed manufacturing processes solidify their market leadership.
- The dominance of adult stem cells in the stem cell therapy market accelerates treatment uptake and investment in regenerative medicine, while enhancing patient access to effective and safe therapies. This leads to improved clinical outcomes and contributes to the future growth of the global stem cell industry.
North America Leads Global Stem Cell Therapy Market Demand
-
North America leads the stem cell therapy market, because of North America healthcare systems have developed strong systems, and regenerative medicine has invested heavily in stem cell therapy development and adoption. for instance, in 2024 the U.S. FDA approved Remestemcel-L, a pediatric acute graft-versus-host disease treatment by Mesoblast Ltd. This fact highlights the high-quality regulatory and innovation environment.
- Additionally, the high rates of the diseases such as cardiovascular disorders, orthopedic injuries, and autoimmune diseases drive demands of stem cell-based therapies. For instance, the MACI cartilage repair therapy by Vericel Corporation is still expanding its market share in the U.S., where it is aimed at treating cartilage defects of the knee in an increasing number of patients. This increased demand will ensure market expansion, increased take up of the therapy and increased dominance of North America in the world leading stem cell therapy market.
- Innovation, patient access, and regional market power in North America are driven by the advanced healthcare system, favorable regulatory framework, and elevated disease burden, factors that significantly influence the development and direction of the stem cell therapy market..
Stem-Cell-Therapy-Market Ecosystem
The global stem cell therapy market is characterized by moderate fragmentation, with a high concentration of key players including Astellas Pharma Inc., Takeda Pharmaceutical Company Limited, Mesoblast Ltd, Vericel Corporation, and Fate Therapeutics Inc. These companies dominate the market due to their strong commercialization capabilities, extensive research and development efforts, regulatory approvals, innovative product pipelines, and high concentration, enabling them to lead the market and promote growth in various therapeutic areas.
The presence of key players enhances market stability, accelerates innovation, and broadens access to higher-order stem cell therapies, contributing to the development of the global stem cell therapy industry and impacting market competition.

Recent Development and Strategic Overview:
-
In August 2025, Fate Therapeutics progressed its FT819 iPSC‑derived off‑the‑shelf CAR T‑cell therapy, initiating treatment in the first severe lupus patients without conditioning, and obtained RMAT designation from the FDA, marking a significant advancement in the development of stem cell–based cellular immunotherapies for autoimmune disorders.
- In December 2024, Takeda updated its strategy to withdraw EU marketing authorization for Alofisel (darvadstrocel), its allogeneic MSC therapy for complex perianal fistulas in Crohn’s disease, reflecting a shift in portfolio focus and search for external partners to advance related cell‑therapy platforms.
Report Scope
|
Detail |
|
|
Market Size in 2025 |
USD 0.6 Bn |
|
Market Forecast Value in 2035 |
~USD 3 Bn |
|
Growth Rate (CAGR) |
17.4% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
US$ Billion for Value |
|
Report Format |
Electronic (PDF) + Excel |
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Stem-Cell-Therapy-Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Stem Cell Therapy Market, By Cell Source |
|
|
Stem Cell Therapy Market, By Cell Type |
|
|
Stem Cell Therapy Market, By Technology |
|
|
Stem Cell Therapy Market, By Therapeutic Application |
|
|
Stem Cell Therapy Market, By Treatment Approach |
|
|
Stem Cell Therapy Market, By End-Use
|
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Stem Cell Therapy Market Outlook
- 2.1.1. Stem Cell Therapy Market Size (Value - US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Stem Cell Therapy Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 3.1.1. Healthcare & Pharmaceutical Ecosystem Analysis
- 3.1.2. Key Trends for Healthcare & Pharmaceutical Industry
- 3.1.3. Regional Distribution for Healthcare & Pharmaceutical Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.4.1. Import & Export Analysis, 2025
- 3.4.2. Top Importing Countries
- 3.4.3. Top Exporting Countries
- 3.5. Trump Tariff Impact Analysis
- 3.5.1. Manufacturer
- 3.5.1.1. Based on the component & Raw material
- 3.5.2. Supply Chain
- 3.5.3. End Consumer
- 3.5.1. Manufacturer
- 3.6. Raw Material Analysis
- 3.1. Global Healthcare & Pharmaceutical Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising prevalence of chronic and degenerative diseases (e.g., cardiovascular, diabetes, neurological disorders)
- 4.1.1.2. Advancements in stem cell research and technologies (including iPSCs and 3D bioprinting)
- 4.1.1.3. Increased clinical trials, regulatory approvals, and supportive government funding
- 4.1.2. Restraints
- 4.1.2.1. High cost of treatment and associated clinical processes
- 4.1.2.2. Regulatory challenges and ethical concerns, especially around embryonic stem cells
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.4.1. Technology & Equipment Suppliers
- 4.4.2. Raw Material & Consumables Suppliers
- 4.4.3. Cell Processing and Manufacturing Companies
- 4.4.4. Clinical Research Organizations and Healthcare Providers
- 4.4.5. Logistics & Commercialization
- 4.5. Porter’s Five Forces Analysis
- 4.6. PESTEL Analysis
- 4.7. Global Stem Cell Therapy Market Demand
- 4.7.1. Historical Market Size – Value (US$ Bn), 2020-2024
- 4.7.2. Current and Future Market Size – Value (US$ Bn), 2026–2035
- 4.7.2.1. Y-o-Y Growth Trends
- 4.7.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Stem Cell Therapy Market Analysis, by Cell Source
- 6.1. Key Segment Analysis
- 6.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Cell Source, 2021-2035
- 6.2.1. Autologous Stem Cells
- 6.2.2. Allogeneic Stem Cells
- 6.2.3. Syngeneic Stem Cells
- 7. Global Stem Cell Therapy Market Analysis, by Cell Type
- 7.1. Key Segment Analysis
- 7.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Cell Type, 2021-2035
- 7.2.1. Adult Stem Cells
- 7.2.1.1. Hematopoietic Stem Cells
- 7.2.1.2. Mesenchymal Stem Cells
- 7.2.1.3. Neural Stem Cells
- 7.2.1.4. Epithelial Stem Cells
- 7.2.2. Embryonic Stem Cells
- 7.2.3. Induced Pluripotent Stem Cells (iPSCs)
- 7.2.4. Very Small Embryonic-Like Stem Cells
- 7.2.1. Adult Stem Cells
- 8. Global Stem Cell Therapy Market Analysis, by Technology
- 8.1. Key Segment Analysis
- 8.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Technology, 2021-2035
- 8.2.1. Cell Acquisition
- 8.2.2. Cell Production
- 8.2.3. Cryopreservation
- 8.2.4. Expansion and Sub-culture
- 9. Global Stem Cell Therapy Market Analysis, by Therapeutic Application
- 9.1. Key Segment Analysis
- 9.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Therapeutic Application, 2021-2035
- 9.2.1. Musculoskeletal Disorders
- 9.2.1.1. Osteoarthritis
- 9.2.1.2. Rheumatoid Arthritis
- 9.2.1.3. Spinal Cord Injury
- 9.2.2. Wounds and Injuries
- 9.2.3. Cardiovascular Diseases
- 9.2.3.1. Myocardial Infarction
- 9.2.3.2. Congestive Heart Failure
- 9.2.4. Inflammatory and Autoimmune Diseases
- 9.2.4.1. Crohn's Disease
- 9.2.4.2. Multiple Sclerosis
- 9.2.5. Neurological Disorders
- 9.2.5.1. Parkinson's Disease
- 9.2.5.2. Alzheimer's Disease
- 9.2.5.3. Stroke
- 9.2.6. Cancer
- 9.2.6.1. Leukemia
- 9.2.6.2. Lymphoma
- 9.2.6.3. Multiple Myeloma
- 9.2.7. Diabetes
- 9.2.7.1. Type 1 Diabetes
- 9.2.7.2. Type 2 Diabetes
- 9.2.8. Liver Disorders
- 9.2.9. Blood Disorders
- 9.2.10. Others
- 9.2.1. Musculoskeletal Disorders
- 10. Global Stem Cell Therapy Market Analysis, by Treatment Approach
- 10.1. Key Segment Analysis
- 10.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Treatment Approach, 2021-2035
- 10.2.1. Stem Cell Transplantation
- 10.2.2. Bone Marrow Transplantation
- 10.2.3. Peripheral Blood Stem Cell Transplantation
- 10.2.4. Cord Blood Transplantation
- 10.2.5. Stem Cell Banking
- 10.2.6. Regenerative Medicine
- 11. Global Stem Cell Therapy Market Analysis, by End-Use
- 11.1. Key Segment Analysis
- 11.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by End-Use, 2021-2035
- 11.2.1. Hospitals & Surgical Centers
- 11.2.2. Specialty Clinics
- 11.2.3. Research Institutes & Academic Centers
- 11.2.4. Pharmaceutical & Biotechnology Companies
- 11.2.5. Cell & Tissue Banks
- 11.2.6. Ambulatory Surgical Centers
- 11.2.7. Others
- 12. Global Stem Cell Therapy Market Analysis and Forecasts, by Region
- 12.1. Key Findings
- 12.2. Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 12.2.1. North America
- 12.2.2. Europe
- 12.2.3. Asia Pacific
- 12.2.4. Middle East
- 12.2.5. Africa
- 12.2.6. South America
- 13. North America Stem Cell Therapy Market Analysis
- 13.1. Key Segment Analysis
- 13.2. Regional Snapshot
- 13.3. North America Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 13.3.1. Cell Source
- 13.3.2. Cell Type
- 13.3.3. Technology
- 13.3.4. Therapeutic Application
- 13.3.5. Treatment Approach
- 13.3.6. End-Use
- 13.3.7. Country
- 13.3.7.1. USA
- 13.3.7.2. Canada
- 13.3.7.3. Mexico
- 13.4. USA Stem Cell Therapy Market
- 13.4.1. Country Segmental Analysis
- 13.4.2. Cell Source
- 13.4.3. Cell Type
- 13.4.4. Technology
- 13.4.5. Therapeutic Application
- 13.4.6. Treatment Approach
- 13.4.7. End-Use
- 13.5. Canada Stem Cell Therapy Market
- 13.5.1. Country Segmental Analysis
- 13.5.2. Cell Source
- 13.5.3. Cell Type
- 13.5.4. Technology
- 13.5.5. Therapeutic Application
- 13.5.6. Treatment Approach
- 13.5.7. End-Use
- 13.6. Mexico Stem Cell Therapy Market
- 13.6.1. Country Segmental Analysis
- 13.6.2. Cell Source
- 13.6.3. Cell Type
- 13.6.4. Technology
- 13.6.5. Therapeutic Application
- 13.6.6. Treatment Approach
- 13.6.7. End-Use
- 14. Europe Stem Cell Therapy Market Analysis
- 14.1. Key Segment Analysis
- 14.2. Regional Snapshot
- 14.3. Europe Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 14.3.1. Cell Source
- 14.3.2. Cell Type
- 14.3.3. Technology
- 14.3.4. Therapeutic Application
- 14.3.5. Treatment Approach
- 14.3.6. End-Use
- 14.3.7. Country
- 14.3.7.1. Germany
- 14.3.7.2. United Kingdom
- 14.3.7.3. France
- 14.3.7.4. Italy
- 14.3.7.5. Spain
- 14.3.7.6. Netherlands
- 14.3.7.7. Nordic Countries
- 14.3.7.8. Poland
- 14.3.7.9. Russia & CIS
- 14.3.7.10. Rest of Europe
- 14.4. Germany Stem Cell Therapy Market
- 14.4.1. Country Segmental Analysis
- 14.4.2. Cell Source
- 14.4.3. Cell Type
- 14.4.4. Technology
- 14.4.5. Therapeutic Application
- 14.4.6. Treatment Approach
- 14.4.7. End-Use
- 14.5. United Kingdom Stem Cell Therapy Market
- 14.5.1. Country Segmental Analysis
- 14.5.2. Cell Source
- 14.5.3. Cell Type
- 14.5.4. Technology
- 14.5.5. Therapeutic Application
- 14.5.6. Treatment Approach
- 14.5.7. End-Use
- 14.6. France Stem Cell Therapy Market
- 14.6.1. Country Segmental Analysis
- 14.6.2. Cell Source
- 14.6.3. Cell Type
- 14.6.4. Technology
- 14.6.5. Therapeutic Application
- 14.6.6. Treatment Approach
- 14.6.7. End-Use
- 14.7. Italy Stem Cell Therapy Market
- 14.7.1. Country Segmental Analysis
- 14.7.2. Cell Source
- 14.7.3. Cell Type
- 14.7.4. Technology
- 14.7.5. Therapeutic Application
- 14.7.6. Treatment Approach
- 14.7.7. End-Use
- 14.8. Spain Stem Cell Therapy Market
- 14.8.1. Country Segmental Analysis
- 14.8.2. Cell Source
- 14.8.3. Cell Type
- 14.8.4. Technology
- 14.8.5. Therapeutic Application
- 14.8.6. Treatment Approach
- 14.8.7. End-Use
- 14.9. Netherlands Stem Cell Therapy Market
- 14.9.1. Country Segmental Analysis
- 14.9.2. Cell Source
- 14.9.3. Cell Type
- 14.9.4. Technology
- 14.9.5. Therapeutic Application
- 14.9.6. Treatment Approach
- 14.9.7. End-Use
- 14.10. Nordic Countries Stem Cell Therapy Market
- 14.10.1. Country Segmental Analysis
- 14.10.2. Cell Source
- 14.10.3. Cell Type
- 14.10.4. Technology
- 14.10.5. Therapeutic Application
- 14.10.6. Treatment Approach
- 14.10.7. End-Use
- 14.11. Poland Stem Cell Therapy Market
- 14.11.1. Country Segmental Analysis
- 14.11.2. Cell Source
- 14.11.3. Cell Type
- 14.11.4. Technology
- 14.11.5. Therapeutic Application
- 14.11.6. Treatment Approach
- 14.11.7. End-Use
- 14.12. Russia & CIS Stem Cell Therapy Market
- 14.12.1. Country Segmental Analysis
- 14.12.2. Cell Source
- 14.12.3. Cell Type
- 14.12.4. Technology
- 14.12.5. Therapeutic Application
- 14.12.6. Treatment Approach
- 14.12.7. End-Use
- 14.13. Rest of Europe Stem Cell Therapy Market
- 14.13.1. Country Segmental Analysis
- 14.13.2. Cell Source
- 14.13.3. Cell Type
- 14.13.4. Technology
- 14.13.5. Therapeutic Application
- 14.13.6. Treatment Approach
- 14.13.7. End-Use
- 15. Asia Pacific Stem Cell Therapy Market Analysis
- 15.1. Key Segment Analysis
- 15.2. Regional Snapshot
- 15.3. Asia Pacific Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 15.3.1. Cell Source
- 15.3.2. Cell Type
- 15.3.3. Technology
- 15.3.4. Therapeutic Application
- 15.3.5. Treatment Approach
- 15.3.6. End-Use
- 15.3.7. Country
- 15.3.7.1. China
- 15.3.7.2. India
- 15.3.7.3. Japan
- 15.3.7.4. South Korea
- 15.3.7.5. Australia and New Zealand
- 15.3.7.6. Indonesia
- 15.3.7.7. Malaysia
- 15.3.7.8. Thailand
- 15.3.7.9. Vietnam
- 15.3.7.10. Rest of Asia Pacific
- 15.4. China Stem Cell Therapy Market
- 15.4.1. Country Segmental Analysis
- 15.4.2. Cell Source
- 15.4.3. Cell Type
- 15.4.4. Technology
- 15.4.5. Therapeutic Application
- 15.4.6. Treatment Approach
- 15.4.7. End-Use
- 15.5. India Stem Cell Therapy Market
- 15.5.1. Country Segmental Analysis
- 15.5.2. Cell Source
- 15.5.3. Cell Type
- 15.5.4. Technology
- 15.5.5. Therapeutic Application
- 15.5.6. Treatment Approach
- 15.5.7. End-Use
- 15.6. Japan Stem Cell Therapy Market
- 15.6.1. Country Segmental Analysis
- 15.6.2. Cell Source
- 15.6.3. Cell Type
- 15.6.4. Technology
- 15.6.5. Therapeutic Application
- 15.6.6. Treatment Approach
- 15.6.7. End-Use
- 15.7. South Korea Stem Cell Therapy Market
- 15.7.1. Country Segmental Analysis
- 15.7.2. Cell Source
- 15.7.3. Cell Type
- 15.7.4. Technology
- 15.7.5. Therapeutic Application
- 15.7.6. Treatment Approach
- 15.7.7. End-Use
- 15.8. Australia and New Zealand Stem Cell Therapy Market
- 15.8.1. Country Segmental Analysis
- 15.8.2. Cell Source
- 15.8.3. Cell Type
- 15.8.4. Technology
- 15.8.5. Therapeutic Application
- 15.8.6. Treatment Approach
- 15.8.7. End-Use
- 15.9. Indonesia Stem Cell Therapy Market
- 15.9.1. Country Segmental Analysis
- 15.9.2. Cell Source
- 15.9.3. Cell Type
- 15.9.4. Technology
- 15.9.5. Therapeutic Application
- 15.9.6. Treatment Approach
- 15.9.7. End-Use
- 15.10. Malaysia Stem Cell Therapy Market
- 15.10.1. Country Segmental Analysis
- 15.10.2. Cell Source
- 15.10.3. Cell Type
- 15.10.4. Technology
- 15.10.5. Therapeutic Application
- 15.10.6. Treatment Approach
- 15.10.7. End-Use
- 15.11. Thailand Stem Cell Therapy Market
- 15.11.1. Country Segmental Analysis
- 15.11.2. Cell Source
- 15.11.3. Cell Type
- 15.11.4. Technology
- 15.11.5. Therapeutic Application
- 15.11.6. Treatment Approach
- 15.11.7. End-Use
- 15.12. Vietnam Stem Cell Therapy Market
- 15.12.1. Country Segmental Analysis
- 15.12.2. Cell Source
- 15.12.3. Cell Type
- 15.12.4. Technology
- 15.12.5. Therapeutic Application
- 15.12.6. Treatment Approach
- 15.12.7. End-Use
- 15.13. Rest of Asia Pacific Stem Cell Therapy Market
- 15.13.1. Country Segmental Analysis
- 15.13.2. Cell Source
- 15.13.3. Cell Type
- 15.13.4. Technology
- 15.13.5. Therapeutic Application
- 15.13.6. Treatment Approach
- 15.13.7. End-Use
- 16. Middle East Stem Cell Therapy Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. Middle East Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Cell Source
- 16.3.2. Cell Type
- 16.3.3. Technology
- 16.3.4. Therapeutic Application
- 16.3.5. Treatment Approach
- 16.3.6. End-Use
- 16.3.7. Country
- 16.3.7.1. Turkey
- 16.3.7.2. UAE
- 16.3.7.3. Saudi Arabia
- 16.3.7.4. Israel
- 16.3.7.5. Rest of Middle East
- 16.4. Turkey Stem Cell Therapy Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Cell Source
- 16.4.3. Cell Type
- 16.4.4. Technology
- 16.4.5. Therapeutic Application
- 16.4.6. Treatment Approach
- 16.4.7. End-Use
- 16.5. UAE Stem Cell Therapy Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Cell Source
- 16.5.3. Cell Type
- 16.5.4. Technology
- 16.5.5. Therapeutic Application
- 16.5.6. Treatment Approach
- 16.5.7. End-Use
- 16.6. Saudi Arabia Stem Cell Therapy Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Cell Source
- 16.6.3. Cell Type
- 16.6.4. Technology
- 16.6.5. Therapeutic Application
- 16.6.6. Treatment Approach
- 16.6.7. End-Use
- 16.7. Israel Stem Cell Therapy Market
- 16.7.1. Country Segmental Analysis
- 16.7.2. Cell Source
- 16.7.3. Cell Type
- 16.7.4. Technology
- 16.7.5. Therapeutic Application
- 16.7.6. Treatment Approach
- 16.7.7. End-Use
- 16.8. Rest of Middle East Stem Cell Therapy Market
- 16.8.1. Country Segmental Analysis
- 16.8.2. Cell Source
- 16.8.3. Cell Type
- 16.8.4. Technology
- 16.8.5. Therapeutic Application
- 16.8.6. Treatment Approach
- 16.8.7. End-Use
- 17. Africa Stem Cell Therapy Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Africa Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Cell Source
- 17.3.2. Cell Type
- 17.3.3. Technology
- 17.3.4. Therapeutic Application
- 17.3.5. Treatment Approach
- 17.3.6. End-Use
- 17.3.7. Country
- 17.3.7.1. South Africa
- 17.3.7.2. Egypt
- 17.3.7.3. Nigeria
- 17.3.7.4. Algeria
- 17.3.7.5. Rest of Africa
- 17.4. South Africa Stem Cell Therapy Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Cell Source
- 17.4.3. Cell Type
- 17.4.4. Technology
- 17.4.5. Therapeutic Application
- 17.4.6. Treatment Approach
- 17.4.7. End-Use
- 17.5. Egypt Stem Cell Therapy Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Cell Source
- 17.5.3. Cell Type
- 17.5.4. Technology
- 17.5.5. Therapeutic Application
- 17.5.6. Treatment Approach
- 17.5.7. End-Use
- 17.6. Nigeria Stem Cell Therapy Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Cell Source
- 17.6.3. Cell Type
- 17.6.4. Technology
- 17.6.5. Therapeutic Application
- 17.6.6. Treatment Approach
- 17.6.7. End-Use
- 17.7. Algeria Stem Cell Therapy Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Cell Source
- 17.7.3. Cell Type
- 17.7.4. Technology
- 17.7.5. Therapeutic Application
- 17.7.6. Treatment Approach
- 17.7.7. End-Use
- 17.8. Rest of Africa Stem Cell Therapy Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Cell Source
- 17.8.3. Cell Type
- 17.8.4. Technology
- 17.8.5. Therapeutic Application
- 17.8.6. Treatment Approach
- 17.8.7. End-Use
- 18. South America Stem Cell Therapy Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. South America Stem Cell Therapy Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Cell Source
- 18.3.2. Cell Type
- 18.3.3. Technology
- 18.3.4. Therapeutic Application
- 18.3.5. Treatment Approach
- 18.3.6. End-Use
- 18.3.7. Country
- 18.3.7.1. Brazil
- 18.3.7.2. Argentina
- 18.3.7.3. Rest of South America
- 18.4. Brazil Stem Cell Therapy Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Cell Source
- 18.4.3. Cell Type
- 18.4.4. Technology
- 18.4.5. Therapeutic Application
- 18.4.6. Treatment Approach
- 18.4.7. End-Use
- 18.5. Argentina Stem Cell Therapy Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Cell Source
- 18.5.3. Cell Type
- 18.5.4. Technology
- 18.5.5. Therapeutic Application
- 18.5.6. Treatment Approach
- 18.5.7. End-Use
- 18.6. Rest of South America Stem Cell Therapy Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Cell Source
- 18.6.3. Cell Type
- 18.6.4. Technology
- 18.6.5. Therapeutic Application
- 18.6.6. Treatment Approach
- 18.6.7. End-Use
- 19. Key Players/ Company Profile
- 19.1. Anterogen Co., Ltd.
- 19.1.1. Company Details/ Overview
- 19.1.2. Company Financials
- 19.1.3. Key Customers and Competitors
- 19.1.4. Business/ Industry Portfolio
- 19.1.5. Product Portfolio/ Specification Details
- 19.1.6. Pricing Data
- 19.1.7. Strategic Overview
- 19.1.8. Recent Developments
- 19.2. Astellas Pharma Inc.
- 19.3. Athersys Inc.
- 19.4. Brainstorm Cell Therapeutics
- 19.5. Caladrius Biosciences Inc.
- 19.6. CBR Systems Inc.
- 19.7. Cellular Engineering Technologies Inc.
- 19.8. CORESTEM Inc.
- 19.9. Cynata Therapeutics Limited
- 19.10. Fate Therapeutics Inc.
- 19.11. Holostem Terapie Avanzate S.r.l.
- 19.12. JCR Pharmaceuticals Co., Ltd.
- 19.13. MEDIPOST Co., Ltd.
- 19.14. Mesoblast Ltd
- 19.15. NuVasive Inc.
- 19.16. Osiris Therapeutics (Smith & Nephew)
- 19.17. Pharmicell Co., Ltd.
- 19.18. Pluristem Therapeutics Inc.
- 19.19. Stempeutics Research Pvt Ltd
- 19.20. Takeda Pharmaceutical Company Limited
- 19.21. Vericel Corporation
- 19.22. ViaCyte Inc.
- 19.23. Other Key Players
- 19.1. Anterogen Co., Ltd.
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
Research Design
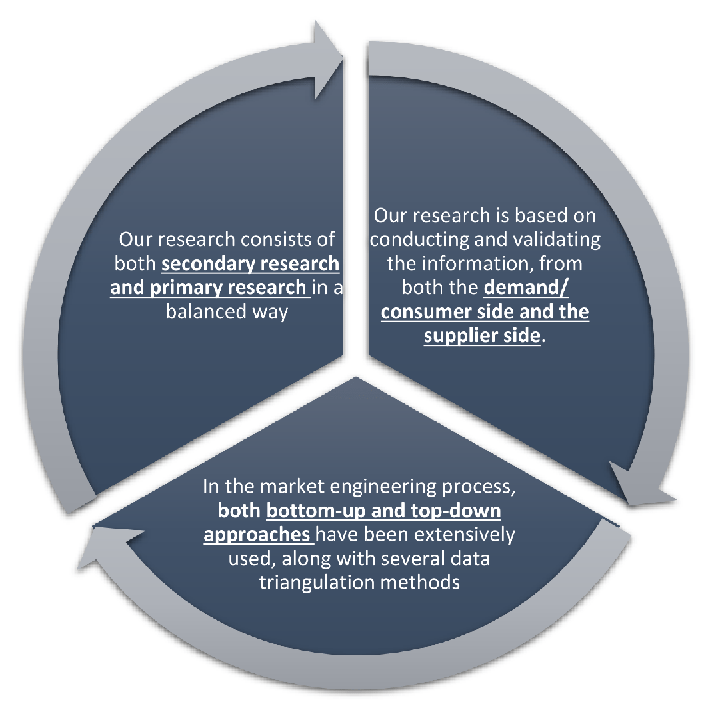
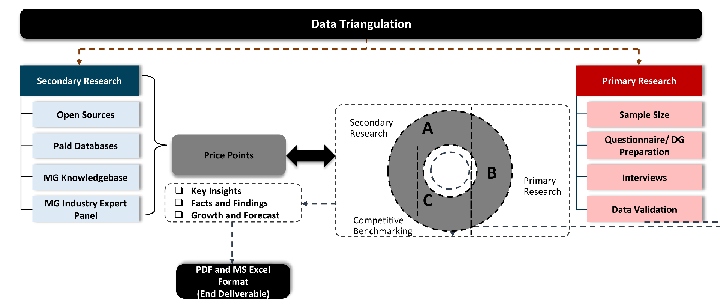
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
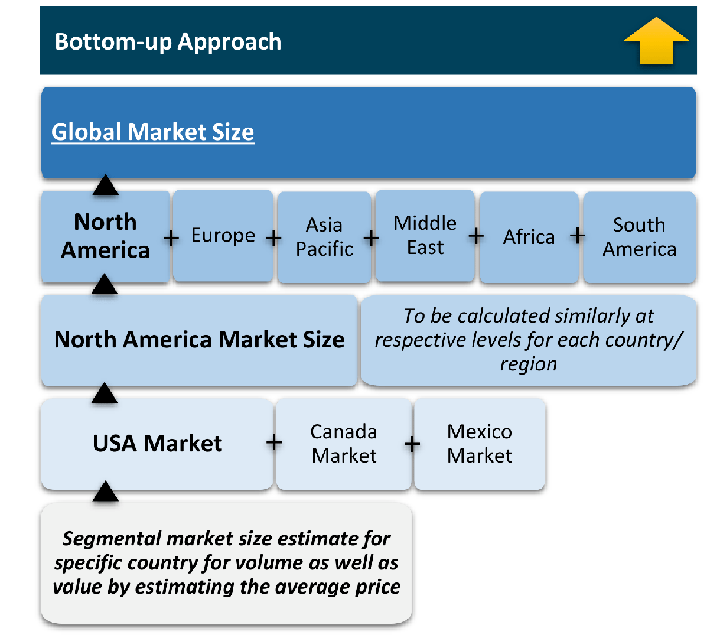
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
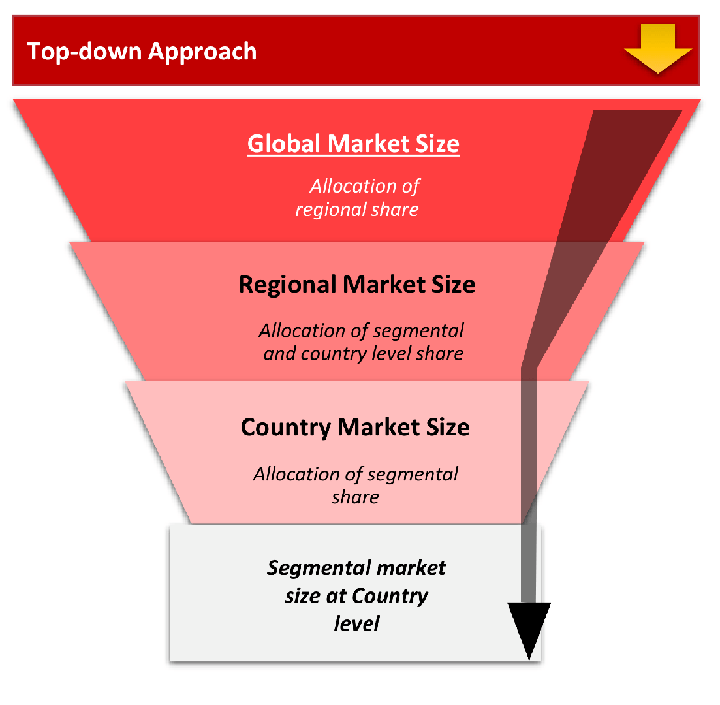
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation