Sterile Barrier Packaging Market Size, Share & Trends Analysis Report by Material Type (Polymers, Paper & Paperboard, Non-woven Fabrics, Foils & Films, Tyvek, Others), Packaging Type, Sterilization Method Compatibility, Barrier Properties, Seal Strength/Type, Package Size/Capacity, Product Format/Structure, Printing & Labeling, End-use Industry, and Geography (North America, Europe, Asia Pacific, Middle East, Africa, and South America) – Global Industry Data, Trends, and Forecasts, 2026–2035
|
|
|
Segmental Data Insights |
|
|
Demand Trends |
|
|
Competitive Landscape |
|
|
Strategic Development |
|
|
Future Outlook & Opportunities |
|
Sterile Barrier Packaging Market Size, Share, and Growth
The global sterile barrier packaging market is experiencing robust growth, with its estimated value of USD 27.7 billion in the year 2025 and USD 50.6 billion by the period 2035, registering a CAGR of 6.2%, during the forecast period. The sterile barrier packaging market is growing rapidly with manufacturers and regulators focusing on the idea of sustainable, recyclable, and circular packaging. The increasing need of bio-based, high-barrier, and compostable materials will guarantee sterility, the safety of products, and the gain of the long shelf-life and the minimization of the influence on the environment.

Felix Neidhart, Sales Manager, Medipack, said, Eastman’s molecular recycling technology makes it possible for us to use certified recycled content without compromising quality or safety ,an essential requirement in medical packaging. It enables the benefits of recycled content in sterile barrier packaging, which was previously not feasible. This is a major step toward circularity in medical packaging.
The sterile barrier packaging market is more and more guided by the two-fold requirements of the regulatory compliance and operational excellence but not as an instrument of high positioning or brand differentiation. The increasing consumer demands regarding product safety, traceability and environmentally friendly packaging are influencing manufacturing companies to re-engineer trays, pouches, peelable films, and rigid or flexible structures to enhance the sterilization assurance, barrier performance, and environmental performance.
The new innovation focus targets the integration of sustainability and functional performance. Companies are adopting bio-based polymers, high barrier mono-material films, recycled PET, and compostable laminates to create packaging that offers a high level of security while reducing the ecological footprint. In September 2025, Olympus Corporation collaborated with DuPont to implement Tyvek with Renewable Attribution to package more than 100 different types of single-use medical devices with high standards of sterility and reduced carbon footprint and sustainable materials sourcing. The implementation of fill-and-seal machines that are compatible with automation, in-line quality control, and uniform practice of production measures all guarantee uniformity and efficiency in the global supply-chain.
Adjacent opportunities to the sterile barrier packaging market include advanced medical device packaging films, pharmaceutical blister and vial systems, aseptic food and beverage packaging, cleanroom-compatible materials, and smart packaging with sterility indicators, leveraging contamination control and regulatory compliance needs, thereby expanding healthcare and life-science adoption while strengthening value beyond traditional medical packaging applications.

Sterile Barrier Packaging Market Dynamics and Trends
Driver: Rising Demand from Healthcare and Pharmaceutical Sectors
-
The rapid evolution of the healthcare products, biologics, pre-filled syringes, implantables, cold-chain pharmaceuticals, etc., is leading into the necessity of high-performance sterile barrier packaging. Hospitals, the clinics and outpatient care providers need packaging with a high level of sterility, long shelf-life, and integrity in transportation, and this aspect is driving a high growth in the market.
- Pharmaceutical companies are also spending more on superior barrier films, peelable pouches, and thermoformed trays that are in conformity with the strict FDA, USP, and ISO 11607 requirements. For instance, in June 2025, the opening of TekniPlex Healthcare, a 200,000-sq-ft flagship in Madison, Wisconsin, to focus on sterile barrier packaging and barrier protection system, increasing medical device and pharmaceutical client capacity.
- The trends along with regulatory control on patient safety and quality assurance support long-term expansion of the sterile barrier packaging market.
Restraint: High Cost and Regulatory & Validation Burdens
-
One of the most significant limitations to the sterile barrier packaging market is that advanced barrier polymers and biodegradable films and medical grade recycled plastics are very expensive. Their materials demand special compounding, accuracy thermoforming, and multi-layers barrier finishes, which has a significant cost of production and capital investment, which affects profitability, especially in small and medium-sized manufacturers.
- The difficulty is enhanced by fluctuation of raw material supply. Post-consumer recycled polymers of high quality, certified bio-based resin, and sustainably obtained paperboard may be irregular because of the gaps in collection between regions, availability seasonally, and increasing regulatory limitations, and therefore procurement becomes unpredictable and more complicated to plan production.
- Manufacturers incur additional cost requirements in the form of strict regulatory compliance, validation testing, and sterilization assurances and logistics of sensitive, temperature-controlled or sterile products.
Opportunity: Adoption of Sustainable and Smart Sterile Packaging Solutions
-
The sterile barrier packaging market is being offered with the prospects of implementing green, recyclable, and computerized packaging structures in medical, pharmaceutical, and biopharma. Increased regulatory interest on material traceability, green polymers, and low carbon footprint, and the interest of healthcare providers in green operations, is causing the demand of materials with both barrier-integrity and recyclability or compostability.
- Manufacturers are using this opportunity by launching new products. For instnace, in October 2025, Evercare introduced Endurance R50, a sterile barrier film, made with up to 50% post-consumer recycled content, and lidded with Tyvek to retain sterilization and barrier properties while reducing carbon footprint, and thus sustainable and high-performance sterile packaging solutions are commercially viable.
- Growing adoption of eco-friendly sterile packaging by hospitals, outpatient centers, and home-care providers is creating opportunities for companies to expand portfolios with smart, multifunctional, and sustainable barrier solutions.
Key Trend: Broadening Use‑Cases beyond Traditional Medical Devices (Biologics, Pre-Filled, Cold Chain, Biopharma)
-
The sterile barrier packaging market is quickly moving beyond the traditional surgical tools and one-use device combinations prompted by the increase of biologics, pre-filled syringes, ready-to-use vials, vaccines as well as cold-chain therapeutics. The market is being remodeled by rising pressure on barrier formats, which guarantee sterilization, stability and cold-chain resiliency throughout manufacturing, storage and distribution chains.
- Special barrier systems have been more intensified with regard to innovation. For instnace, in September 2025, TekniPlex Healthcare has developed a sterile barrier packaging offering that also incorporates the use of barrier-blister films and custom liquid-drug-delivery systems that will be utilized in pharmaceutical, biologic, and injectable therapies as an illustration of the way companies are now moving out of traditional device packaging into broader biopharma and cold-chain use-cases.
- The rising use of biologics and advanced therapies is increasing demand for sterile packaging beyond hospitals, including home care, outpatient use, and distribution of prefilled syringes, auto-injectors, and temperature-sensitive therapies.
Sterile-Barrier-Packaging-Market Analysis and Segmental Data
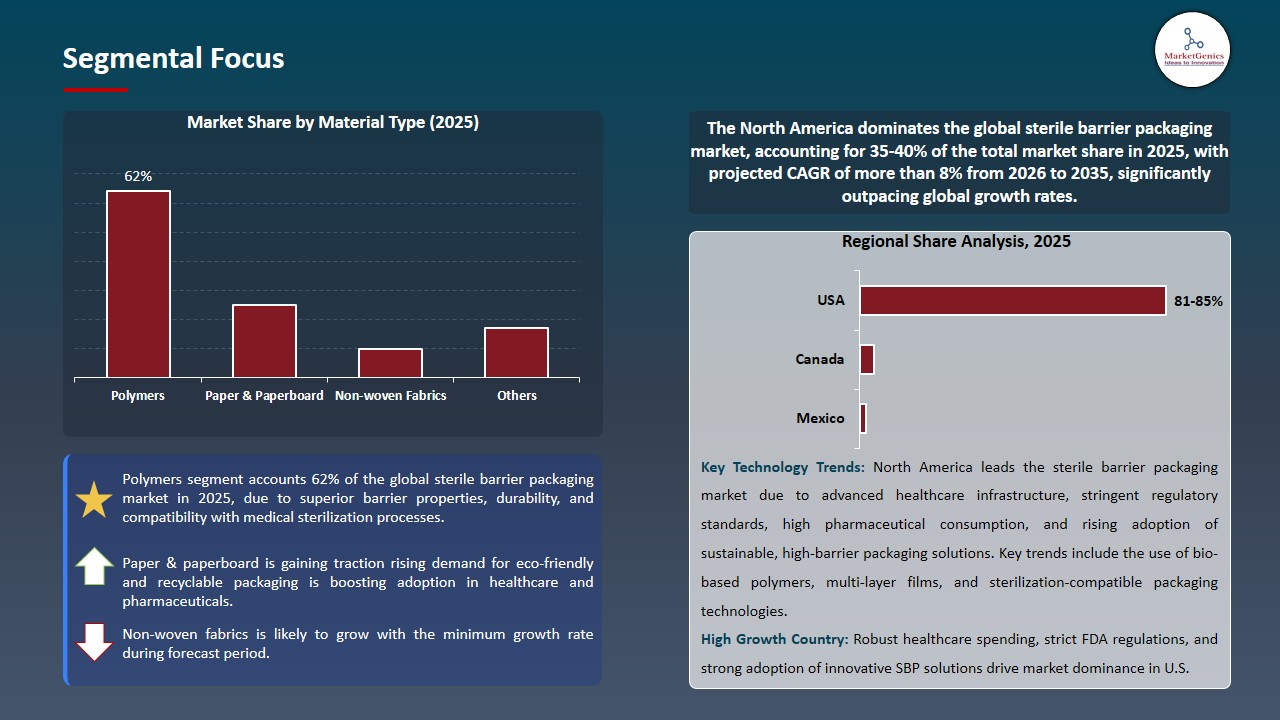
Polymers Dominate Global Sterile Barrier Packaging Market
-
High-performance polymers dominates the sterile barrier packaging market because they can be reliably sterilized, their validity is microbial-barrier performance, and they are in accord with FDA, USP, and ISO 11607. Their high chemical stability, capability to resist steam, EO and gamma sterilization and light weight properties allow manufacturers to provide safe, sterile and durable packaging of surgical equipment and diagnostic kits as well as implantable equipment.
- There is an increasing trend in innovation of recycle and bio-based polymers. For instance, in 2023, Eastar Renew 6763 copolyester started being used by Pacur, LLC, to manufacture rigid, thermoformed sterile barrier medical-device packaging, with sterility protection and up to 50 percent recycled content.
- Their consistency in processing, barrier performance and support of sophisticated sterilization techniques continues to make them supreme in sterile packaging in health care and life sciences industries.
North America Leads Global Sterile Barrier Packaging Market Demand
-
North America is dominating the global sterile barrier packaging market with the high degree of sterility guaranteed by strict FDA, USP and ISO 11607 standards and partially by confirmed barrier performance and total trackability over the medical and pharmaceutical supply chains. High-volume of surgical procedures, high standards of healthcare facilities in the region, and quick uptake of single-use sterile devices are some of the factors that have ensured a constant need in the high-performance sterile packaging formats.
- Manufacturers in North America are rolling out medical grade barrier films, breathable sterilization substrates and high strength peelable systems that are compatible with steam, EO and radiation sterilization. The adaption of Eastar Renew 6763 recycled-content copolyester by Nelipak Healthcare to make rigid thermoformed sterile barrier packaging of Class II / Class III medical equipment, which combines sterility consideration with environmental sustainability.
- The rising use of biologics and advanced therapies is increasing demand for sterile packaging beyond hospitals, including home care, outpatient use, and distribution of prefilled syringes, auto-injectors, and temperature-sensitive therapies.
Sterile-Barrier-Packaging-Market Ecosystem
The global sterile barrier packaging market is fairly fragmented, with a combination of multinational material innovators, medical grade packaging specialists and local converters and suppliers serving healthcare, pharmaceutical, diagnostics and life-science packaging. The leading companies in Tier-1 including Amcor plc, DuPont (Tyvek), and 3M Company are leading by their rich portfolio of sterile-grade materials, international manufacturing presence, and high-level of R&D in high-barrier materials, microbial-resistant structures, and materials that can be sterilized. Different suppliers such as West Pharmaceutical Services and Sealed Air Corporation (Tier-2) provide specialized formats like sterile pouches, trays, thermoformed systems as well as barrier films, mostly used by device manufacturers and supply-chain intermediaries demanding validated sterility assurance levels (SAL).
The ecosystem incorporates suppliers of raw-material of medical polymers, specialty nonwovens and high-performance films, converters that are operating in cleanroom production, sterilization service providers, OEMs, and hospital procurement channels. To enhance the performance of sterility, compliance, and competitiveness in the market, investments are getting more focused on automation, digital traceability, materials that represent sustainability, lifecycle validation, and regulatory harmonization.

Recent Development and Strategic Overview
-
In June 2025, the Healthmark Industries released their SafeGuard Dry Sterilization Wrap which was designed to avoid wet-pack failures by having two layers that consist of hydrophobic SMS outer layer and a hydrophilic inner layer. The wrap improves the drying performance, preserves microbial-barrier, and facilitates the uniform sterile-processing results.
- In November 2025, Medipack introduced its new thermoformed barrier film, MEDPETG 6763 RN, with Eastar 6763 Renew, a copolymer that has a maximum of 50% recycled content. The film maintains the clarity, retention and sterilization needed in sterile medical packaging to perform both high-performance barrier functionality and reduced environmental footprint.
Report Scope
|
Detail |
|
|
Market Size in 2025 |
USD 27.7 Bn |
|
Market Forecast Value in 2035 |
USD 50.6 Bn |
|
Growth Rate (CAGR) |
6.2% |
|
Forecast Period |
2026 – 2035 |
|
Historical Data Available for |
2021 – 2024 |
|
Market Size Units |
US$ Billion for Value |
|
Report Format |
Electronic (PDF) + Excel |
|
North America |
Europe |
Asia Pacific |
Middle East |
Africa |
South America |
|
|
|
|
|
|
|
Companies Covered |
|||||
|
|
|
|
|
|
Sterile-Barrier-Packaging-Market Segmentation and Highlights
|
Segment |
Sub-segment |
|
Sterile Barrier Packaging Market, By Material Type |
|
|
Sterile Barrier Packaging Market, By Packaging Type |
|
|
Sterile Barrier Packaging Market, By Sterilization Method Compatibility |
|
|
Sterile Barrier Packaging Market, By Barrier Properties |
|
|
Sterile Barrier Packaging Market, By Seal Strength/Type |
|
|
Sterile Barrier Packaging Market, By Package Size/Capacity |
|
|
Sterile Barrier Packaging Market, By Product Format/Structure |
|
|
Sterile Barrier Packaging Market, By Printing & Labeling |
|
|
Sterile Barrier Packaging Market, By End-use Industry |
|
Frequently Asked Questions
Table of Contents
- 1. Research Methodology and Assumptions
- 1.1. Definitions
- 1.2. Research Design and Approach
- 1.3. Data Collection Methods
- 1.4. Base Estimates and Calculations
- 1.5. Forecasting Models
- 1.5.1. Key Forecast Factors & Impact Analysis
- 1.6. Secondary Research
- 1.6.1. Open Sources
- 1.6.2. Paid Databases
- 1.6.3. Associations
- 1.7. Primary Research
- 1.7.1. Primary Sources
- 1.7.2. Primary Interviews with Stakeholders across Ecosystem
- 2. Executive Summary
- 2.1. Global Sterile Barrier Packaging Market Outlook
- 2.1.1. Sterile Barrier Packaging Market Size (Value - US$ Bn), and Forecasts, 2021-2035
- 2.1.2. Compounded Annual Growth Rate Analysis
- 2.1.3. Growth Opportunity Analysis
- 2.1.4. Segmental Share Analysis
- 2.1.5. Geographical Share Analysis
- 2.2. Market Analysis and Facts
- 2.3. Supply-Demand Analysis
- 2.4. Competitive Benchmarking
- 2.5. Go-to- Market Strategy
- 2.5.1. Customer/ End-use Industry Assessment
- 2.5.2. Growth Opportunity Data, 2026-2035
- 2.5.2.1. Regional Data
- 2.5.2.2. Country Data
- 2.5.2.3. Segmental Data
- 2.5.3. Identification of Potential Market Spaces
- 2.5.4. GAP Analysis
- 2.5.5. Potential Attractive Price Points
- 2.5.6. Prevailing Market Risks & Challenges
- 2.5.7. Preferred Sales & Marketing Strategies
- 2.5.8. Key Recommendations and Analysis
- 2.5.9. A Way Forward
- 2.1. Global Sterile Barrier Packaging Market Outlook
- 3. Industry Data and Premium Insights
- 3.1. Global Packaging Industry Overview, 2025
- 3.1.1. Packaging Industry Ecosystem Analysis
- 3.1.2. Key Trends for Packaging Industry
- 3.1.3. Regional Distribution for Packaging Industry
- 3.2. Supplier Customer Data
- 3.3. Technology Roadmap and Developments
- 3.4. Trade Analysis
- 3.4.1. Import & Export Analysis, 2025
- 3.4.2. Top Importing Countries
- 3.4.3. Top Exporting Countries
- 3.5. Trump Tariff Impact Analysis
- 3.5.1. Manufacturer
- 3.5.1.1. Based on the component & Raw material
- 3.5.2. Supply Chain
- 3.5.3. End Consumer
- 3.5.1. Manufacturer
- 3.6. Raw Material Analysis
- 3.1. Global Packaging Industry Overview, 2025
- 4. Market Overview
- 4.1. Market Dynamics
- 4.1.1. Drivers
- 4.1.1.1. Rising demand from healthcare and pharmaceutical sectors for safe, contamination-free packaging.
- 4.1.1.2. Increasing regulatory and compliance requirements for sterile and high-barrier packaging.
- 4.1.1.3. Growing adoption of sustainable and circular packaging solutions in medical and food applications.
- 4.1.2. Restraints
- 4.1.2.1. High production and raw material costs, including specialized equipment and barrier materials.
- 4.1.2.2. Complex regulatory validation and quality assurance processes limiting adoption, especially in emerging markets.
- 4.1.1. Drivers
- 4.2. Key Trend Analysis
- 4.3. Regulatory Framework
- 4.3.1. Key Regulations, Norms, and Subsidies, by Key Countries
- 4.3.2. Tariffs and Standards
- 4.3.3. Impact Analysis of Regulations on the Market
- 4.4. Value Chain Analysis
- 4.4.1. Raw Material Suppliers
- 4.4.2. Manufacturers
- 4.4.3. Dealers/ Distributors
- 4.4.4. End-Users/ Customers
- 4.5. Porter’s Five Forces Analysis
- 4.6. PESTEL Analysis
- 4.7. Global Sterile Barrier Packaging Market Demand
- 4.7.1. Historical Market Size – Value (US$ Bn), 2020-2024
- 4.7.2. Current and Future Market Size – Value (US$ Bn), 2026–2035
- 4.7.2.1. Y-o-Y Growth Trends
- 4.7.2.2. Absolute $ Opportunity Assessment
- 4.1. Market Dynamics
- 5. Competition Landscape
- 5.1. Competition structure
- 5.1.1. Fragmented v/s consolidated
- 5.2. Company Share Analysis, 2025
- 5.2.1. Global Company Market Share
- 5.2.2. By Region
- 5.2.2.1. North America
- 5.2.2.2. Europe
- 5.2.2.3. Asia Pacific
- 5.2.2.4. Middle East
- 5.2.2.5. Africa
- 5.2.2.6. South America
- 5.3. Product Comparison Matrix
- 5.3.1. Specifications
- 5.3.2. Market Positioning
- 5.3.3. Pricing
- 5.1. Competition structure
- 6. Global Sterile Barrier Packaging Market Analysis, by Material Type
- 6.1. Key Segment Analysis
- 6.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Material Type, 2021-2035
- 6.2.1. Polymers
- 6.2.1.1. Polyethylene (PE)
- 6.2.1.2. Polypropylene (PP)
- 6.2.1.3. Polyethylene Terephthalate (PET)
- 6.2.1.4. Polyvinyl Chloride (PVC)
- 6.2.1.5. Polyvinylidene Chloride (PVDC)
- 6.2.1.6. High-Density Polyethylene (HDPE)
- 6.2.1.7. Low-Density Polyethylene (LDPE)
- 6.2.1.8. Others
- 6.2.2. Paper & Paperboard
- 6.2.2.1. Medical Grade Paper
- 6.2.2.2. Coated Paper
- 6.2.2.3. Crepe Paper
- 6.2.2.4. Kraft Paper
- 6.2.2.5. Others
- 6.2.3. Non-woven Fabrics
- 6.2.3.1. Spunbond
- 6.2.3.2. Meltblown
- 6.2.3.3. SMS (Spunbond-Meltblown-Spunbond)
- 6.2.3.4. Others
- 6.2.4. Foils & Films
- 6.2.4.1. Aluminum Foil
- 6.2.4.2. Multi-layer Films
- 6.2.4.3. Breathable Films
- 6.2.4.4. Others
- 6.2.5. Tyvek
- 6.2.6. Others
- 6.2.1. Polymers
- 7. Global Sterile Barrier Packaging Market Analysis, by Packaging Type
- 7.1. Key Segment Analysis
- 7.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Packaging Type, 2021-2035
- 7.2.1. Pouches & Bags
- 7.2.1.1. Flat Pouches
- 7.2.1.2. Gusseted Pouches
- 7.2.1.3. Stand-up Pouches
- 7.2.1.4. Header Bags
- 7.2.1.5. Others
- 7.2.2. Thermoformed Trays
- 7.2.2.1. Shallow Trays
- 7.2.2.2. Deep Trays
- 7.2.2.3. Compartment Trays
- 7.2.2.4. Others
- 7.2.3. Lidding Films & Foils
- 7.2.4. Reels & Rolls
- 7.2.5. Wraps
- 7.2.6. Clamshell Packaging
- 7.2.7. Blister Packaging
- 7.2.8. Others
- 7.2.1. Pouches & Bags
- 8. Global Sterile Barrier Packaging Market Analysis, by Sterilization Method Compatibility
- 8.1. Key Segment Analysis
- 8.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Sterilization Method Compatibility, 2021-2035
- 8.2.1. Ethylene Oxide (ETO) Sterilization
- 8.2.2. Gamma Radiation Sterilization
- 8.2.3. E-Beam Radiation Sterilization
- 8.2.4. Steam/Autoclave Sterilization
- 8.2.5. Hydrogen Peroxide Sterilization
- 8.2.6. Formaldehyde Sterilization
- 8.2.7. Multiple Sterilization Compatible
- 9. Global Sterile Barrier Packaging Market Analysis, by Barrier Properties
- 9.1. Key Segment Analysis
- 9.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Barrier Properties, 2021-2035
- 9.2.1. High Barrier
- 9.2.1.1. Oxygen Barrier
- 9.2.1.2. Moisture Barrier
- 9.2.1.3. Light Barrier
- 9.2.1.4. Microbial Barrier
- 9.2.1.5. Others
- 9.2.2. Medium Barrier
- 9.2.3. Low Barrier
- 9.2.4. Breathable Barrier
- 9.2.1. High Barrier
- 10. Global Sterile Barrier Packaging Market Analysis, by Seal Strength/Type
- 10.1. Key Segment Analysis
- 10.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Seal Strength/Type, 2021-2035
- 10.2.1. Peelable Seals
- 10.2.1.1. Easy Peel
- 10.2.1.2. Standard Peel
- 10.2.1.3. Controlled Peel
- 10.2.1.4. Others
- 10.2.2. Heat Seals
- 10.2.3. Cold Seals
- 10.2.4. Pressure-Sensitive Seals
- 10.2.5. Ultrasonic Seals
- 10.2.6. High-Strength Seals
- 10.2.1. Peelable Seals
- 11. Global Sterile Barrier Packaging Market Analysis, by Package Size/Capacity
- 11.1. Key Segment Analysis
- 11.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Package Size/Capacity, 2021-2035
- 11.2.1. Small (< 100 sq cm)
- 11.2.2. Medium (100-500 sq cm)
- 11.2.3. Large (500-1000 sq cm)
- 11.2.4. Extra Large (> 1000 sq cm)
- 12. Global Sterile Barrier Packaging Market Analysis, by Product Format/Structure
- 12.1. Key Segment Analysis
- 12.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Product Format/Structure, 2021-2035
- 12.2.1. Single Layer
- 12.2.2. Multi-Layer
- 12.2.3. Coated
- 12.2.4. Laminated
- 12.2.5. Co-extruded
- 13. Global Sterile Barrier Packaging Market Analysis, by Printing & Labeling
- 13.1. Key Segment Analysis
- 13.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Printing & Labeling, 2021-2035
- 13.2.1. Pre-printed Packaging
- 13.2.2. Plain/Unprinted Packaging
- 13.2.3. Direct Printable Surface
- 13.2.4. Label Ready
- 13.2.5. Color-coded Packaging
- 14. Global Sterile Barrier Packaging Market Analysis, by End-use Industry
- 14.1. Key Segment Analysis
- 14.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Package Size/Capacity, 2021-2035
- 14.2.1. Medical Devices Industry
- 14.2.1.1. Surgical Instruments Packaging
- 14.2.1.2. Implantable Devices Packaging
- 14.2.1.3. Diagnostic Equipment Packaging
- 14.2.1.4. Dental Instruments Packaging
- 14.2.1.5. Ophthalmic Devices Packaging
- 14.2.1.6. Cardiovascular Devices Packaging
- 14.2.1.7. Orthopedic Devices Packaging
- 14.2.1.8. Catheters & Tubing Packaging
- 14.2.1.9. Others
- 14.2.2. Pharmaceutical Industry
- 14.2.2.1. Pre-filled Syringes Packaging
- 14.2.2.2. Vials & Ampoules Packaging
- 14.2.2.3. Injectable Drug Packaging
- 14.2.2.4. Tablet & Capsule Blister Packaging
- 14.2.2.5. Medical Kits Packaging
- 14.2.2.6. IV Sets Packaging
- 14.2.2.7. Drug Delivery Devices Packaging
- 14.2.2.8. Others
- 14.2.3. Hospital & Healthcare Facilities
- 14.2.3.1. Surgical Kits & Trays Packaging
- 14.2.3.2. Wound Care Products Packaging
- 14.2.3.3. IV Administration Sets Packaging
- 14.2.3.4. Dialysis Products Packaging
- 14.2.3.5. Operating Room Supplies Packaging
- 14.2.3.6. Emergency Medical Supplies Packaging
- 14.2.3.7. Others
- 14.2.4. In-Vitro Diagnostics (IVD)
- 14.2.4.1. Test Kits Packaging
- 14.2.4.2. Laboratory Reagents Packaging
- 14.2.4.3. Sample Collection Devices Packaging
- 14.2.4.4. Diagnostic Strips Packaging
- 14.2.4.5. Microplates Packaging
- 14.2.4.6. Others
- 14.2.5. Biotechnology & Life Sciences
- 14.2.5.1. Cell Culture Products Packaging
- 14.2.5.2. Biological Samples Packaging
- 14.2.5.3. Research Instruments Packaging
- 14.2.5.4. Lab Consumables Packaging
- 14.2.5.5. Others
- 14.2.6. Medical Textile & Wipes Industry
- 14.2.6.1. Surgical Gowns Packaging
- 14.2.6.2. Drapes & Covers Packaging
- 14.2.6.3. Medical Wipes Packaging
- 14.2.6.4. Sterile Gauze & Dressings Packaging
- 14.2.6.5. Others
- 14.2.7. Contract Manufacturing Organizations (CMOs)
- 14.2.7.1. Custom Medical Device Packaging
- 14.2.7.2. Contract Sterilization Packaging
- 14.2.7.3. Third-party Manufacturing Packaging
- 14.2.7.4. Others
- 14.2.8. Veterinary Medicine
- 14.2.8.1. Veterinary Surgical Instruments Packaging
- 14.2.8.2. Animal Healthcare Products Packaging
- 14.2.8.3. Veterinary Diagnostic Products Packaging
- 14.2.8.4. Others
- 14.2.1. Medical Devices Industry
- 15. Global Sterile Barrier Packaging Market Analysis and Forecasts, by Region
- 15.1. Key Findings
- 15.2. Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, by Region, 2021-2035
- 15.2.1. North America
- 15.2.2. Europe
- 15.2.3. Asia Pacific
- 15.2.4. Middle East
- 15.2.5. Africa
- 15.2.6. South America
- 16. North America Sterile Barrier Packaging Market Analysis
- 16.1. Key Segment Analysis
- 16.2. Regional Snapshot
- 16.3. North America Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 16.3.1. Material Type
- 16.3.2. Packaging Type
- 16.3.3. Sterilization Method Compatibility
- 16.3.4. Barrier Properties
- 16.3.5. Seal Strength/Type
- 16.3.6. Package Size/Capacity
- 16.3.7. Product Format/Structure
- 16.3.8. Printing & Labeling
- 16.3.9. End-use Industry
- 16.3.10. Country
- 16.3.10.1. USA
- 16.3.10.2. Canada
- 16.3.10.3. Mexico
- 16.4. USA Sterile Barrier Packaging Market
- 16.4.1. Country Segmental Analysis
- 16.4.2. Material Type
- 16.4.3. Packaging Type
- 16.4.4. Sterilization Method Compatibility
- 16.4.5. Barrier Properties
- 16.4.6. Seal Strength/Type
- 16.4.7. Package Size/Capacity
- 16.4.8. Product Format/Structure
- 16.4.9. Printing & Labeling
- 16.4.10. End-use Industry
- 16.5. Canada Sterile Barrier Packaging Market
- 16.5.1. Country Segmental Analysis
- 16.5.2. Material Type
- 16.5.3. Packaging Type
- 16.5.4. Sterilization Method Compatibility
- 16.5.5. Barrier Properties
- 16.5.6. Seal Strength/Type
- 16.5.7. Package Size/Capacity
- 16.5.8. Product Format/Structure
- 16.5.9. Printing & Labeling
- 16.5.10. End-use Industry
- 16.6. Mexico Sterile Barrier Packaging Market
- 16.6.1. Country Segmental Analysis
- 16.6.2. Material Type
- 16.6.3. Packaging Type
- 16.6.4. Sterilization Method Compatibility
- 16.6.5. Barrier Properties
- 16.6.6. Seal Strength/Type
- 16.6.7. Package Size/Capacity
- 16.6.8. Product Format/Structure
- 16.6.9. Printing & Labeling
- 16.6.10. End-use Industry
- 17. Europe Sterile Barrier Packaging Market Analysis
- 17.1. Key Segment Analysis
- 17.2. Regional Snapshot
- 17.3. Europe Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 17.3.1. Material Type
- 17.3.2. Packaging Type
- 17.3.3. Sterilization Method Compatibility
- 17.3.4. Barrier Properties
- 17.3.5. Seal Strength/Type
- 17.3.6. Package Size/Capacity
- 17.3.7. Product Format/Structure
- 17.3.8. Printing & Labeling
- 17.3.9. End-use Industry
- 17.3.10. Country
- 17.3.10.1. Germany
- 17.3.10.2. United Kingdom
- 17.3.10.3. France
- 17.3.10.4. Italy
- 17.3.10.5. Spain
- 17.3.10.6. Netherlands
- 17.3.10.7. Nordic Countries
- 17.3.10.8. Poland
- 17.3.10.9. Russia & CIS
- 17.3.10.10. Rest of Europe
- 17.4. Germany Sterile Barrier Packaging Market
- 17.4.1. Country Segmental Analysis
- 17.4.2. Material Type
- 17.4.3. Packaging Type
- 17.4.4. Sterilization Method Compatibility
- 17.4.5. Barrier Properties
- 17.4.6. Seal Strength/Type
- 17.4.7. Package Size/Capacity
- 17.4.8. Product Format/Structure
- 17.4.9. Printing & Labeling
- 17.4.10. End-use Industry
- 17.5. United Kingdom Sterile Barrier Packaging Market
- 17.5.1. Country Segmental Analysis
- 17.5.2. Material Type
- 17.5.3. Packaging Type
- 17.5.4. Sterilization Method Compatibility
- 17.5.5. Barrier Properties
- 17.5.6. Seal Strength/Type
- 17.5.7. Package Size/Capacity
- 17.5.8. Product Format/Structure
- 17.5.9. Printing & Labeling
- 17.5.10. End-use Industry
- 17.6. France Sterile Barrier Packaging Market
- 17.6.1. Country Segmental Analysis
- 17.6.2. Material Type
- 17.6.3. Packaging Type
- 17.6.4. Sterilization Method Compatibility
- 17.6.5. Barrier Properties
- 17.6.6. Seal Strength/Type
- 17.6.7. Package Size/Capacity
- 17.6.8. Product Format/Structure
- 17.6.9. Printing & Labeling
- 17.6.10. End-use Industry
- 17.7. Italy Sterile Barrier Packaging Market
- 17.7.1. Country Segmental Analysis
- 17.7.2. Material Type
- 17.7.3. Packaging Type
- 17.7.4. Sterilization Method Compatibility
- 17.7.5. Barrier Properties
- 17.7.6. Seal Strength/Type
- 17.7.7. Package Size/Capacity
- 17.7.8. Product Format/Structure
- 17.7.9. Printing & Labeling
- 17.7.10. End-use Industry
- 17.8. Spain Sterile Barrier Packaging Market
- 17.8.1. Country Segmental Analysis
- 17.8.2. Material Type
- 17.8.3. Packaging Type
- 17.8.4. Sterilization Method Compatibility
- 17.8.5. Barrier Properties
- 17.8.6. Seal Strength/Type
- 17.8.7. Package Size/Capacity
- 17.8.8. Product Format/Structure
- 17.8.9. Printing & Labeling
- 17.8.10. End-use Industry
- 17.9. Netherlands Sterile Barrier Packaging Market
- 17.9.1. Country Segmental Analysis
- 17.9.2. Material Type
- 17.9.3. Packaging Type
- 17.9.4. Sterilization Method Compatibility
- 17.9.5. Barrier Properties
- 17.9.6. Seal Strength/Type
- 17.9.7. Package Size/Capacity
- 17.9.8. Product Format/Structure
- 17.9.9. Printing & Labeling
- 17.9.10. End-use Industry
- 17.10. Nordic Countries Sterile Barrier Packaging Market
- 17.10.1. Country Segmental Analysis
- 17.10.2. Material Type
- 17.10.3. Packaging Type
- 17.10.4. Sterilization Method Compatibility
- 17.10.5. Barrier Properties
- 17.10.6. Seal Strength/Type
- 17.10.7. Package Size/Capacity
- 17.10.8. Product Format/Structure
- 17.10.9. Printing & Labeling
- 17.10.10. End-use Industry
- 17.11. Poland Sterile Barrier Packaging Market
- 17.11.1. Country Segmental Analysis
- 17.11.2. Material Type
- 17.11.3. Packaging Type
- 17.11.4. Sterilization Method Compatibility
- 17.11.5. Barrier Properties
- 17.11.6. Seal Strength/Type
- 17.11.7. Package Size/Capacity
- 17.11.8. Product Format/Structure
- 17.11.9. Printing & Labeling
- 17.11.10. End-use Industry
- 17.12. Russia & CIS Sterile Barrier Packaging Market
- 17.12.1. Country Segmental Analysis
- 17.12.2. Material Type
- 17.12.3. Packaging Type
- 17.12.4. Sterilization Method Compatibility
- 17.12.5. Barrier Properties
- 17.12.6. Seal Strength/Type
- 17.12.7. Package Size/Capacity
- 17.12.8. Product Format/Structure
- 17.12.9. Printing & Labeling
- 17.12.10. End-use Industry
- 17.13. Rest of Europe Sterile Barrier Packaging Market
- 17.13.1. Country Segmental Analysis
- 17.13.2. Material Type
- 17.13.3. Packaging Type
- 17.13.4. Sterilization Method Compatibility
- 17.13.5. Barrier Properties
- 17.13.6. Seal Strength/Type
- 17.13.7. Package Size/Capacity
- 17.13.8. Product Format/Structure
- 17.13.9. Printing & Labeling
- 17.13.10. End-use Industry
- 18. Asia Pacific Sterile Barrier Packaging Market Analysis
- 18.1. Key Segment Analysis
- 18.2. Regional Snapshot
- 18.3. Asia Pacific Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 18.3.1. Material Type
- 18.3.2. Packaging Type
- 18.3.3. Sterilization Method Compatibility
- 18.3.4. Barrier Properties
- 18.3.5. Seal Strength/Type
- 18.3.6. Package Size/Capacity
- 18.3.7. Product Format/Structure
- 18.3.8. Printing & Labeling
- 18.3.9. End-use Industry
- 18.3.10. Country
- 18.3.10.1. China
- 18.3.10.2. India
- 18.3.10.3. Japan
- 18.3.10.4. South Korea
- 18.3.10.5. Australia and New Zealand
- 18.3.10.6. Indonesia
- 18.3.10.7. Malaysia
- 18.3.10.8. Thailand
- 18.3.10.9. Vietnam
- 18.3.10.10. Rest of Asia Pacific
- 18.4. China Sterile Barrier Packaging Market
- 18.4.1. Country Segmental Analysis
- 18.4.2. Material Type
- 18.4.3. Packaging Type
- 18.4.4. Sterilization Method Compatibility
- 18.4.5. Barrier Properties
- 18.4.6. Seal Strength/Type
- 18.4.7. Package Size/Capacity
- 18.4.8. Product Format/Structure
- 18.4.9. Printing & Labeling
- 18.4.10. End-use Industry
- 18.5. India Sterile Barrier Packaging Market
- 18.5.1. Country Segmental Analysis
- 18.5.2. Material Type
- 18.5.3. Packaging Type
- 18.5.4. Sterilization Method Compatibility
- 18.5.5. Barrier Properties
- 18.5.6. Seal Strength/Type
- 18.5.7. Package Size/Capacity
- 18.5.8. Product Format/Structure
- 18.5.9. Printing & Labeling
- 18.5.10. End-use Industry
- 18.6. Japan Sterile Barrier Packaging Market
- 18.6.1. Country Segmental Analysis
- 18.6.2. Material Type
- 18.6.3. Packaging Type
- 18.6.4. Sterilization Method Compatibility
- 18.6.5. Barrier Properties
- 18.6.6. Seal Strength/Type
- 18.6.7. Package Size/Capacity
- 18.6.8. Product Format/Structure
- 18.6.9. Printing & Labeling
- 18.6.10. End-use Industry
- 18.7. South Korea Sterile Barrier Packaging Market
- 18.7.1. Country Segmental Analysis
- 18.7.2. Material Type
- 18.7.3. Packaging Type
- 18.7.4. Sterilization Method Compatibility
- 18.7.5. Barrier Properties
- 18.7.6. Seal Strength/Type
- 18.7.7. Package Size/Capacity
- 18.7.8. Product Format/Structure
- 18.7.9. Printing & Labeling
- 18.7.10. End-use Industry
- 18.8. Australia and New Zealand Sterile Barrier Packaging Market
- 18.8.1. Country Segmental Analysis
- 18.8.2. Material Type
- 18.8.3. Packaging Type
- 18.8.4. Sterilization Method Compatibility
- 18.8.5. Barrier Properties
- 18.8.6. Seal Strength/Type
- 18.8.7. Package Size/Capacity
- 18.8.8. Product Format/Structure
- 18.8.9. Printing & Labeling
- 18.8.10. End-use Industry
- 18.9. Indonesia Sterile Barrier Packaging Market
- 18.9.1. Country Segmental Analysis
- 18.9.2. Material Type
- 18.9.3. Packaging Type
- 18.9.4. Sterilization Method Compatibility
- 18.9.5. Barrier Properties
- 18.9.6. Seal Strength/Type
- 18.9.7. Package Size/Capacity
- 18.9.8. Product Format/Structure
- 18.9.9. Printing & Labeling
- 18.9.10. End-use Industry
- 18.10. Malaysia Sterile Barrier Packaging Market
- 18.10.1. Country Segmental Analysis
- 18.10.2. Material Type
- 18.10.3. Packaging Type
- 18.10.4. Sterilization Method Compatibility
- 18.10.5. Barrier Properties
- 18.10.6. Seal Strength/Type
- 18.10.7. Package Size/Capacity
- 18.10.8. Product Format/Structure
- 18.10.9. Printing & Labeling
- 18.10.10. End-use Industry
- 18.11. Thailand Sterile Barrier Packaging Market
- 18.11.1. Country Segmental Analysis
- 18.11.2. Material Type
- 18.11.3. Packaging Type
- 18.11.4. Sterilization Method Compatibility
- 18.11.5. Barrier Properties
- 18.11.6. Seal Strength/Type
- 18.11.7. Package Size/Capacity
- 18.11.8. Product Format/Structure
- 18.11.9. Printing & Labeling
- 18.11.10. End-use Industry
- 18.12. Vietnam Sterile Barrier Packaging Market
- 18.12.1. Country Segmental Analysis
- 18.12.2. Material Type
- 18.12.3. Packaging Type
- 18.12.4. Sterilization Method Compatibility
- 18.12.5. Barrier Properties
- 18.12.6. Seal Strength/Type
- 18.12.7. Package Size/Capacity
- 18.12.8. Product Format/Structure
- 18.12.9. Printing & Labeling
- 18.12.10. End-use Industry
- 18.13. Rest of Asia Pacific Sterile Barrier Packaging Market
- 18.13.1. Country Segmental Analysis
- 18.13.2. Material Type
- 18.13.3. Packaging Type
- 18.13.4. Sterilization Method Compatibility
- 18.13.5. Barrier Properties
- 18.13.6. Seal Strength/Type
- 18.13.7. Package Size/Capacity
- 18.13.8. Product Format/Structure
- 18.13.9. Printing & Labeling
- 18.13.10. End-use Industry
- 19. Middle East Sterile Barrier Packaging Market Analysis
- 19.1. Key Segment Analysis
- 19.2. Regional Snapshot
- 19.3. Middle East Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 19.3.1. Material Type
- 19.3.2. Packaging Type
- 19.3.3. Sterilization Method Compatibility
- 19.3.4. Barrier Properties
- 19.3.5. Seal Strength/Type
- 19.3.6. Package Size/Capacity
- 19.3.7. Product Format/Structure
- 19.3.8. Printing & Labeling
- 19.3.9. End-use Industry
- 19.3.10. Country
- 19.3.10.1. Turkey
- 19.3.10.2. UAE
- 19.3.10.3. Saudi Arabia
- 19.3.10.4. Israel
- 19.3.10.5. Rest of Middle East
- 19.4. Turkey Sterile Barrier Packaging Market
- 19.4.1. Country Segmental Analysis
- 19.4.2. Material Type
- 19.4.3. Packaging Type
- 19.4.4. Sterilization Method Compatibility
- 19.4.5. Barrier Properties
- 19.4.6. Seal Strength/Type
- 19.4.7. Package Size/Capacity
- 19.4.8. Product Format/Structure
- 19.4.9. Printing & Labeling
- 19.4.10. End-use Industry
- 19.5. UAE Sterile Barrier Packaging Market
- 19.5.1. Country Segmental Analysis
- 19.5.2. Material Type
- 19.5.3. Packaging Type
- 19.5.4. Sterilization Method Compatibility
- 19.5.5. Barrier Properties
- 19.5.6. Seal Strength/Type
- 19.5.7. Package Size/Capacity
- 19.5.8. Product Format/Structure
- 19.5.9. Printing & Labeling
- 19.5.10. End-use Industry
- 19.6. Saudi Arabia Sterile Barrier Packaging Market
- 19.6.1. Country Segmental Analysis
- 19.6.2. Material Type
- 19.6.3. Packaging Type
- 19.6.4. Sterilization Method Compatibility
- 19.6.5. Barrier Properties
- 19.6.6. Seal Strength/Type
- 19.6.7. Package Size/Capacity
- 19.6.8. Product Format/Structure
- 19.6.9. Printing & Labeling
- 19.6.10. End-use Industry
- 19.7. Israel Sterile Barrier Packaging Market
- 19.7.1. Country Segmental Analysis
- 19.7.2. Material Type
- 19.7.3. Packaging Type
- 19.7.4. Sterilization Method Compatibility
- 19.7.5. Barrier Properties
- 19.7.6. Seal Strength/Type
- 19.7.7. Package Size/Capacity
- 19.7.8. Product Format/Structure
- 19.7.9. Printing & Labeling
- 19.7.10. End-use Industry
- 19.8. Rest of Middle East Sterile Barrier Packaging Market
- 19.8.1. Country Segmental Analysis
- 19.8.2. Material Type
- 19.8.3. Packaging Type
- 19.8.4. Sterilization Method Compatibility
- 19.8.5. Barrier Properties
- 19.8.6. Seal Strength/Type
- 19.8.7. Package Size/Capacity
- 19.8.8. Product Format/Structure
- 19.8.9. Printing & Labeling
- 19.8.10. End-use Industry
- 20. Africa Sterile Barrier Packaging Market Analysis
- 20.1. Key Segment Analysis
- 20.2. Regional Snapshot
- 20.3. Africa Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 20.3.1. Material Type
- 20.3.2. Packaging Type
- 20.3.3. Sterilization Method Compatibility
- 20.3.4. Barrier Properties
- 20.3.5. Seal Strength/Type
- 20.3.6. Package Size/Capacity
- 20.3.7. Product Format/Structure
- 20.3.8. Printing & Labeling
- 20.3.9. End-use Industry
- 20.3.10. Country
- 20.3.10.1. South Africa
- 20.3.10.2. Egypt
- 20.3.10.3. Nigeria
- 20.3.10.4. Algeria
- 20.3.10.5. Rest of Africa
- 20.4. South Africa Sterile Barrier Packaging Market
- 20.4.1. Country Segmental Analysis
- 20.4.2. Material Type
- 20.4.3. Packaging Type
- 20.4.4. Sterilization Method Compatibility
- 20.4.5. Barrier Properties
- 20.4.6. Seal Strength/Type
- 20.4.7. Package Size/Capacity
- 20.4.8. Product Format/Structure
- 20.4.9. Printing & Labeling
- 20.4.10. End-use Industry
- 20.5. Egypt Sterile Barrier Packaging Market
- 20.5.1. Country Segmental Analysis
- 20.5.2. Material Type
- 20.5.3. Packaging Type
- 20.5.4. Sterilization Method Compatibility
- 20.5.5. Barrier Properties
- 20.5.6. Seal Strength/Type
- 20.5.7. Package Size/Capacity
- 20.5.8. Product Format/Structure
- 20.5.9. Printing & Labeling
- 20.5.10. End-use Industry
- 20.6. Nigeria Sterile Barrier Packaging Market
- 20.6.1. Country Segmental Analysis
- 20.6.2. Material Type
- 20.6.3. Packaging Type
- 20.6.4. Sterilization Method Compatibility
- 20.6.5. Barrier Properties
- 20.6.6. Seal Strength/Type
- 20.6.7. Package Size/Capacity
- 20.6.8. Product Format/Structure
- 20.6.9. Printing & Labeling
- 20.6.10. End-use Industry
- 20.7. Algeria Sterile Barrier Packaging Market
- 20.7.1. Country Segmental Analysis
- 20.7.2. Material Type
- 20.7.3. Packaging Type
- 20.7.4. Sterilization Method Compatibility
- 20.7.5. Barrier Properties
- 20.7.6. Seal Strength/Type
- 20.7.7. Package Size/Capacity
- 20.7.8. Product Format/Structure
- 20.7.9. Printing & Labeling
- 20.7.10. End-use Industry
- 20.8. Rest of Africa Sterile Barrier Packaging Market
- 20.8.1. Country Segmental Analysis
- 20.8.2. Material Type
- 20.8.3. Packaging Type
- 20.8.4. Sterilization Method Compatibility
- 20.8.5. Barrier Properties
- 20.8.6. Seal Strength/Type
- 20.8.7. Package Size/Capacity
- 20.8.8. Product Format/Structure
- 20.8.9. Printing & Labeling
- 20.8.10. End-use Industry
- 21. South America Sterile Barrier Packaging Market Analysis
- 21.1. Key Segment Analysis
- 21.2. Regional Snapshot
- 21.3. South America Sterile Barrier Packaging Market Size (Value - US$ Bn), Analysis, and Forecasts, 2021-2035
- 21.3.1. Material Type
- 21.3.2. Packaging Type
- 21.3.3. Sterilization Method Compatibility
- 21.3.4. Barrier Properties
- 21.3.5. Seal Strength/Type
- 21.3.6. Package Size/Capacity
- 21.3.7. Product Format/Structure
- 21.3.8. Printing & Labeling
- 21.3.9. End-use Industry
- 21.3.10. Country
- 21.3.10.1. Brazil
- 21.3.10.2. Argentina
- 21.3.10.3. Rest of South America
- 21.4. Brazil Sterile Barrier Packaging Market
- 21.4.1. Country Segmental Analysis
- 21.4.2. Material Type
- 21.4.3. Packaging Type
- 21.4.4. Sterilization Method Compatibility
- 21.4.5. Barrier Properties
- 21.4.6. Seal Strength/Type
- 21.4.7. Package Size/Capacity
- 21.4.8. Product Format/Structure
- 21.4.9. Printing & Labeling
- 21.4.10. End-use Industry
- 21.5. Argentina Sterile Barrier Packaging Market
- 21.5.1. Country Segmental Analysis
- 21.5.2. Material Type
- 21.5.3. Packaging Type
- 21.5.4. Sterilization Method Compatibility
- 21.5.5. Barrier Properties
- 21.5.6. Seal Strength/Type
- 21.5.7. Package Size/Capacity
- 21.5.8. Product Format/Structure
- 21.5.9. Printing & Labeling
- 21.5.10. End-use Industry
- 21.6. Rest of South America Sterile Barrier Packaging Market
- 21.6.1. Country Segmental Analysis
- 21.6.2. Material Type
- 21.6.3. Packaging Type
- 21.6.4. Sterilization Method Compatibility
- 21.6.5. Barrier Properties
- 21.6.6. Seal Strength/Type
- 21.6.7. Package Size/Capacity
- 21.6.8. Product Format/Structure
- 21.6.9. Printing & Labeling
- 21.6.10. End-use Industry
- 22. Key Players/ Company Profile
- 22.1. 3M Company
- 22.1.1. Company Details/ Overview
- 22.1.2. Company Financials
- 22.1.3. Key Customers and Competitors
- 22.1.4. Business/ Industry Portfolio
- 22.1.5. Product Portfolio/ Specification Details
- 22.1.6. Pricing Data
- 22.1.7. Strategic Overview
- 22.1.8. Recent Developments
- 22.2. Amcor plc
- 22.3. Bemis Company (Amcor)
- 22.4. Berry Global Inc.
- 22.5. Constantia Flexibles Group
- 22.6. DuPont (Tyvek)
- 22.7. Klöckner Pentaplast
- 22.8. Mitsubishi Chemical Holdings
- 22.9. Nelipak Healthcare Packaging
- 22.10. Orchids Paper Products Company
- 22.11. Placon Corporation
- 22.12. Printpack Inc.
- 22.13. ProAmpac
- 22.14. Riverside Medical Packaging Company
- 22.15. Sealed Air Corporation
- 22.16. Sonoco Products Company
- 22.17. Steripack Contract Manufacturing
- 22.18. Technipaq Inc.
- 22.19. UFP Technologies Inc.
- 22.20. West Pharmaceutical Services
- 22.21. Wipak Group (Wihuri Group)
- 22.22. Other Key Players
- 22.1. 3M Company
Note* - This is just tentative list of players. While providing the report, we will cover more number of players based on their revenue and share for each geography
Research Design
Our research design integrates both demand-side and supply-side analysis through a balanced combination of primary and secondary research methodologies. By utilizing both bottom-up and top-down approaches alongside rigorous data triangulation methods, we deliver robust market intelligence that supports strategic decision-making.
MarketGenics' comprehensive research design framework ensures the delivery of accurate, reliable, and actionable market intelligence. Through the integration of multiple research approaches, rigorous validation processes, and expert analysis, we provide our clients with the insights needed to make informed strategic decisions and capitalize on market opportunities.
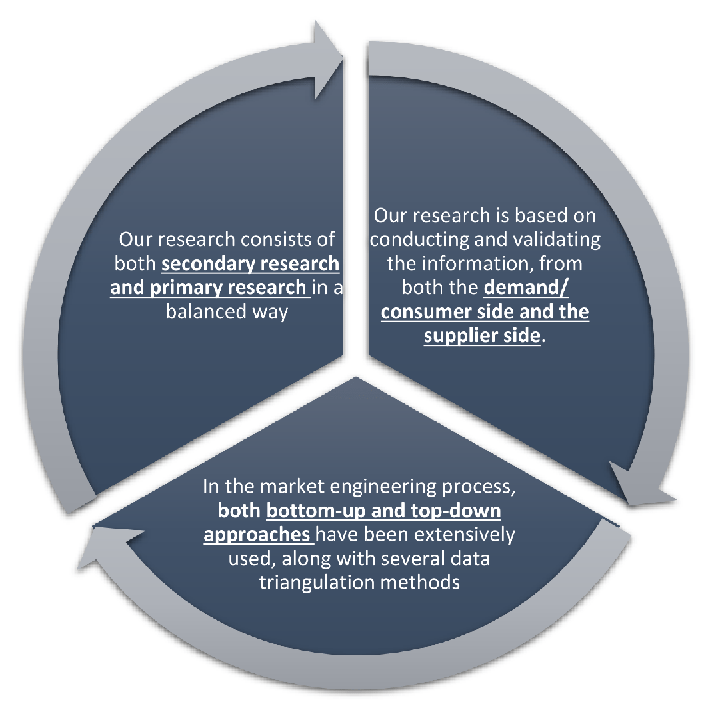
MarketGenics leverages a dedicated industry panel of experts and a comprehensive suite of paid databases to effectively collect, consolidate, and analyze market intelligence.
Our approach has consistently proven to be reliable and effective in generating accurate market insights, identifying key industry trends, and uncovering emerging business opportunities.
Through both primary and secondary research, we capture and analyze critical company-level data such as manufacturing footprints, including technical centers, R&D facilities, sales offices, and headquarters.
Our expert panel further enhances our ability to estimate market size for specific brands based on validated field-level intelligence.
Our data mining techniques incorporate both parametric and non-parametric methods, allowing for structured data collection, sorting, processing, and cleaning.
Demand projections are derived from large-scale data sets analyzed through proprietary algorithms, culminating in robust and reliable market sizing.
Research Approach
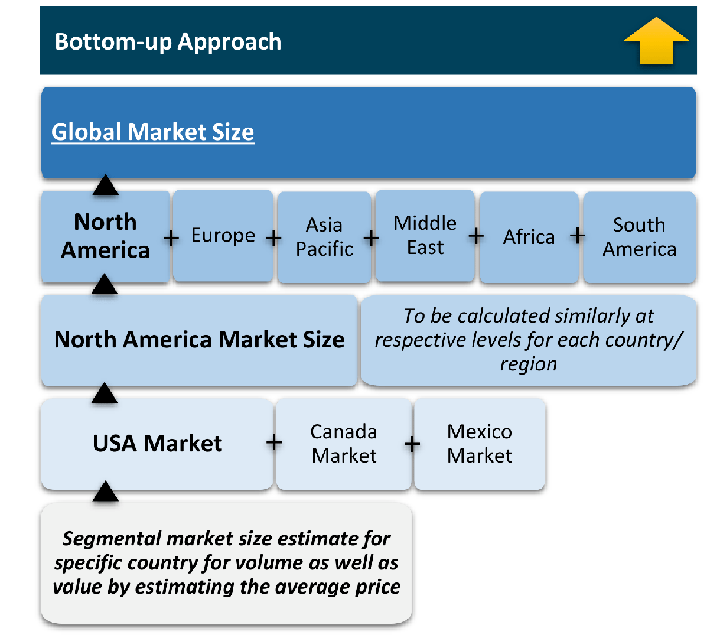
The bottom-up approach builds market estimates by starting with the smallest addressable market units and systematically aggregating them to create comprehensive market size projections.
This method begins with specific, granular data points and builds upward to create the complete market landscape.
Customer Analysis → Segmental Analysis → Geographical Analysis
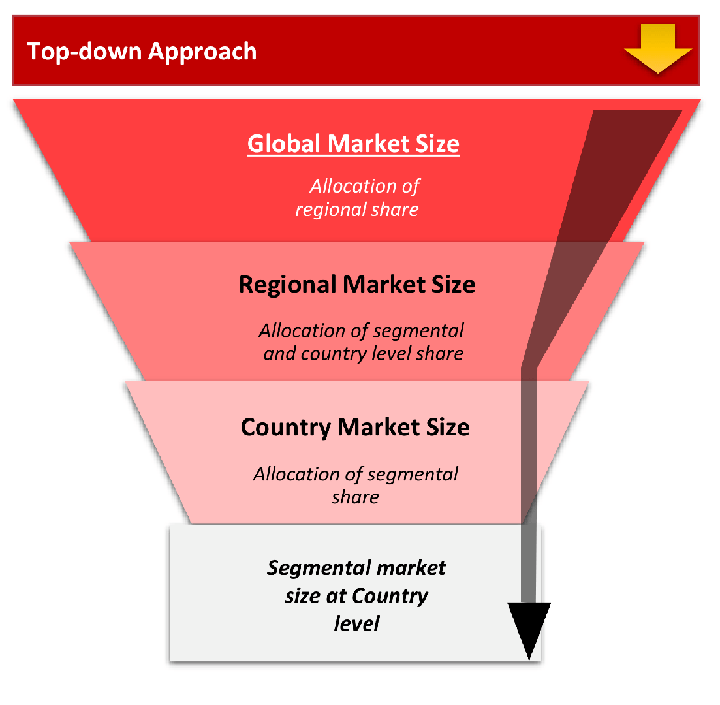
The top-down approach starts with the broadest possible market data and systematically narrows it down through a series of filters and assumptions to arrive at specific market segments or opportunities.
This method begins with the big picture and works downward to increasingly specific market slices.
TAM → SAM → SOM
Research Methods
Desk / Secondary Research
While analysing the market, we extensively study secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and commercial report. Secondary sources that we utilize are not only the public sources, but it is a combination of Open Source, Associations, Paid Databases, MG Repository & Knowledgebase, and others.
- Company websites, annual reports, financial reports, broker reports, and investor presentations
- National government documents, statistical databases and reports
- News articles, press releases and web-casts specific to the companies operating in the market, Magazines, reports, and others
- We gather information from commercial data sources for deriving company specific data such as segmental revenue, share for geography, product revenue, and others
- Internal and external proprietary databases (industry-specific), relevant patent, and regulatory databases
- Governing Bodies, Government Organizations
- Relevant Authorities, Country-specific Associations for Industries
We also employ the model mapping approach to estimate the product level market data through the players' product portfolio
Primary Research
Primary research/ interviews is vital in analyzing the market. Most of the cases involves paid primary interviews. Primary sources include primary interviews through e-mail interactions, telephonic interviews, surveys as well as face-to-face interviews with the different stakeholders across the value chain including several industry experts.
| Type of Respondents | Number of Primaries |
|---|---|
| Tier 2/3 Suppliers | ~20 |
| Tier 1 Suppliers | ~25 |
| End-users | ~25 |
| Industry Expert/ Panel/ Consultant | ~30 |
| Total | ~100 |
MG Knowledgebase
• Repository of industry blog, newsletter and case studies
• Online platform covering detailed market reports, and company profiles
Forecasting Factors and Models
Forecasting Factors
- Historical Trends – Past market patterns, cycles, and major events that shaped how markets behave over time. Understanding past trends helps predict future behavior.
- Industry Factors – Specific characteristics of the industry like structure, regulations, and innovation cycles that affect market dynamics.
- Macroeconomic Factors – Economic conditions like GDP growth, inflation, and employment rates that affect how much money people have to spend.
- Demographic Factors – Population characteristics like age, income, and location that determine who can buy your product.
- Technology Factors – How quickly people adopt new technology and how much technology infrastructure exists.
- Regulatory Factors – Government rules, laws, and policies that can help or restrict market growth.
- Competitive Factors – Analyzing competition structure such as degree of competition and bargaining power of buyers and suppliers.
Forecasting Models / Techniques
Multiple Regression Analysis
- Identify and quantify factors that drive market changes
- Statistical modeling to establish relationships between market drivers and outcomes
Time Series Analysis – Seasonal Patterns
- Understand regular cyclical patterns in market demand
- Advanced statistical techniques to separate trend, seasonal, and irregular components
Time Series Analysis – Trend Analysis
- Identify underlying market growth patterns and momentum
- Statistical analysis of historical data to project future trends
Expert Opinion – Expert Interviews
- Gather deep industry insights and contextual understanding
- In-depth interviews with key industry stakeholders
Multi-Scenario Development
- Prepare for uncertainty by modeling different possible futures
- Creating optimistic, pessimistic, and most likely scenarios
Time Series Analysis – Moving Averages
- Sophisticated forecasting for complex time series data
- Auto-regressive integrated moving average models with seasonal components
Econometric Models
- Apply economic theory to market forecasting
- Sophisticated economic models that account for market interactions
Expert Opinion – Delphi Method
- Harness collective wisdom of industry experts
- Structured, multi-round expert consultation process
Monte Carlo Simulation
- Quantify uncertainty and probability distributions
- Thousands of simulations with varying input parameters
Research Analysis
Our research framework is built upon the fundamental principle of validating market intelligence from both demand and supply perspectives. This dual-sided approach ensures comprehensive market understanding and reduces the risk of single-source bias.
Demand-Side Analysis: We understand end-user/application behavior, preferences, and market needs along with the penetration of the product for specific application.
Supply-Side Analysis: We estimate overall market revenue, analyze the segmental share along with industry capacity, competitive landscape, and market structure.
Validation & Evaluation
Data triangulation is a validation technique that uses multiple methods, sources, or perspectives to examine the same research question, thereby increasing the credibility and reliability of research findings. In market research, triangulation serves as a quality assurance mechanism that helps identify and minimize bias, validate assumptions, and ensure accuracy in market estimates.
- Data Source Triangulation – Using multiple data sources to examine the same phenomenon
- Methodological Triangulation – Using multiple research methods to study the same research question
- Investigator Triangulation – Using multiple researchers or analysts to examine the same data
- Theoretical Triangulation – Using multiple theoretical perspectives to interpret the same data
Custom Market Research Services
We will customise the research for you, in case the report listed above does not meet your requirements.
Get 10% Free Customisation